Which of the following are chiral? H3C H;C H3C CH3 CH3 H3C CH3 CH3 H;C CI H;C
Question:
Which of the following are chiral?
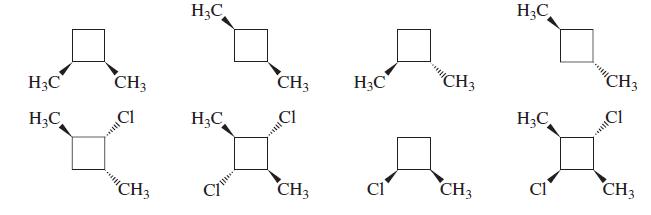
Transcribed Image Text:
H3C H;C H3C CH3 CH3 H3C CH3 "CH3 H;C CI H;C CI H;C CH3 CH3 Cl CH3 CI CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)

Answered By

Salman Ahsan
Did my graduation as well as master's from a reputed central university of India i.e Jamia Millia Islamia in New Delhi. Qualified CSIR-NET (a national level entrance examination for doctorate) in Chemical Sciences, with a national rank of 19. Have been teaching at various institutions from my bachelor's days. Currently working in the field of Ekectroorganic Synthesis at a premier research institute of India i.e. Indian Institute of Science Education and Research Bhopal.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following objects are chiral? (a) Screwdriver (b) Screw (c) Beanstalk (d) Shoe
-
Which of the following are chiral? (a) 2-Methyl-2,3-hexadiene (b) 4-Methyl-2,3-hexadiene (c) 2,4-Dimethyl-2,3-pentadiene
-
Which of the following objects are chiral and which are achiral? a. Golf club b. Teacup c. Football d. Corkscrew e. Tennis racket f. Shoe g. Portrait h. Pencil
-
When members of your team coordinate how to prepare the progress report, in which direction is the flow of communication?
-
Briefly describe how a magnetic sector mass spectrometer works.
-
Determine the force in each member of the truss and state if the members are in tension or compression. Set P1 = 10 kN, P2 = 8 kN. F E 2 m D. A B 2 m P2
-
How would you treat unsatisfactory responses that are discovered in editing?
-
The separate income statements of Danner Company and its 90%-owned subsidiary, Link Company, for the year ended December 31, 2012, are as follows: The following additional facts apply: a. On January...
-
Hildreth Company uses a job order cost system. The following data summarize the operations related to production for April, the first month of operations: Materials purchased on account, $ 3 , 3 7 0...
-
In June, current year, Wendy Winger organized a corporation to provide aerial photography services. The company, called Aerial Views, began operations immediately. Transactions during the month of...
-
Which of the following compounds has a stereoisomer that is a meso compound? a. 2,4-dibromohexane b. 2,4-dibromopentane c. 2,4-dimethylpentane d. 1,3-dichlorocyclohexane e. 1,4-dichlorocyclohexane f....
-
There are two stable isotopes of nitrogen, 14 N and 15 N. How many protons and neutrons does each isotope have?
-
Determine the ionic charge for the metal cation in each of the following compounds: (a) Hg(OH) 2 (b) Co(ClO 3 ) 3 .
-
Explain what is meant by the online value proposition, and give two examples of the value proposition for websites with which you are familiar.
-
What is the role of monitoring in the strategic planning process?
-
Summarise the main tangible and intangible business benefits of the Internet to a company.
-
What is meant by the Internet contribution, and what is its relevance to strategy?
-
Summarise the key elements of the macro-environment that should be scanned by an e-commerce manager.
-
The present value of JECK Co.'s expected free cash flows is $100 million. If JECK has $30 million in debt, $6 million in cash, and 2 million shares outstanding, what is its share price?
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
Which compound should undergo substitution of the bromine by phenolate anion: 4 - bromopyridine or 3 - bromopyridine ? Explain, and give the structure of the product.
-
Outline a synthesis for each of the following compounds from the indicated starting material and any other reagents. 4-methyl-3-nitropyridine from - lpicoline
-
Predict the predominant product in each of the following reactions. Explain your answer. 3,4-dibromopyridine + NH3, heat (C5H5BrN2)
-
Provide References In September of 2015, the EPA issued Volkswagen ( OTCPK:VLKAY ) a notice of violation of the Clean Air Act of 1963. The EPA accused the Volkswagen Group of intentionally...
-
2. Find c such that A is invertible 1 0 A= -1 -1 3 1 0 2c -4]
-
Futura Company purchases the 4 0 , 0 0 0 starters that it installs in its standard line of farm tractors from a supplier for the price of $ 8 . 4 0 per unit. Due to a reduction in output, the company...

Study smarter with the SolutionInn App


