A reaction has yielded a mixture of the following two stereoisomers. Explain how you would use proton
Question:
A reaction has yielded a mixture of the following two stereoisomers. Explain how you would use proton NMR to determine which is which.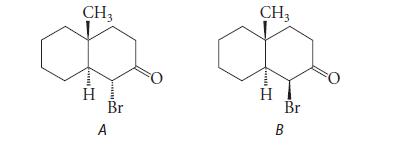
Transcribed Image Text:
CH3 HH. T Br A CH3 H Br B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Both stereoisomers are based on a transdecalinlike structure You could determine which of the stereo...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Tell precisely how you would use the proton NMR spectra to distinguish between the following pairs of compounds. (a) 1-bromopropane and 2-bromopropane (b) (c) (d) CH3-CH-t-CH 3 (CH3)2C -_CH3 and CHCH...
-
Explain how you would use Table V to find the two 2-values that divide the area under a 2-curve into a middle 0.95 area and two outside 0.025 areas. Find these two 2-values for a 2-curve with df = 14.
-
The owner of Colonial Adventure Tours would like you to complete the following tasks to help him maintain his database. In the following exercises, you will use the data in the Colonial Adventure...
-
The combined sewer system in city ABC is comprised of two parallel interceptors referred to as "North" and "South" lines. The southern line is connected to a newly built wastewater treatment plant....
-
Adjustments to the balance per bank Required Identify which of the following items are added to or subtracted from the unadjusted bank balance to arrive at the true cash balance. Distinguish the...
-
In the previous question, over what range of lease payments will the lease be profitable for both parties? You work for a nuclear research laboratory that is contemplating leasing a diagnostic...
-
Purina entered in a contract with the defendant to sell the defendant piglets, known as weanlingsbaby pigs that have been weaned. It is uncontested that the buyer breached and that Purina is entitled...
-
The president of the retailer Prime Products has just approached the companys bank with a request for a $30,000, 90-day loan. The purpose of the loan is to assist the company in acquiring...
-
Two blocks are fastened to the ceiling of an elevator as in Figure. The elevator accelerates upward at 2.00 m/s. Find the tension in each rope. T 10.0 kg T 10.0 kg
-
Outline a synthesis of the following compound from acetylene and any other compounds containing no more than five carbons: CH3(CH)6, CH,CH,CH(CH3)2 C=C H H cis-2-methyl-5-tridecene
-
You work for a reputable chemical supply house. An angry customer, Fly Ofterhandle, has called, alleging that a sample of 2,5-hexanediol he purchased cannot be the correct compound. As evidence, he...
-
Describe the scale of redistribution in the United States.
-
The ZAP Electric Co. manufactures electric circuit breakers. The circuit breakers are produced on two different assembly lines in the company's Spokane plant. Assembly line I is highly automated and...
-
How does the correlation between returns on a project and returns on the firms other assets affect the projects risk? The stand-alone risk of an individual corporate project may be quite high, but...
-
Differentiate between a stocks expected rate of return (r), required rate of return (r), and realized, after-the-fact historical return (r). Which would have to be larger to induce you to buy the...
-
Suppose a nearby concert hall booked a different one of your favorite performers every night for the next month. Make up a table showing your demand schedule for tickets.
-
The effect of tax rate on WACC Rayyan Games, an IT firm, wishes to explore the effect on its cost of capital of the rate at which the company pays taxes. The firm wishes to maintain a capital...
-
Distinguish group term life insurance from whole life insurance.
-
Ashlee, Hiroki, Kate, and Albee LLC each own a 25 percent interest in Tally Industries LLC, which generates annual gross receipts of over $10 million. Ashlee, Hiroki, and Kate manage the business,...
-
Draw the structure and assign Z or F stereochemistry to the product you expect from E2 reaction of the following molecule with NaOH (yellow-green =Cl):
-
Which compound in each of the following pairs will react faster in an SN2 reaction with OH? (a) CH3Br or CH3I (b) CH3CH2I in ethanol or in dimethyl sulfoxide (c) (CH3) 3CC1 or CH3C1 (d) H2C = CHBr or...
-
What effect would you expect the following changes to have on the rate of the SN2 reaction of 1-iodo-2-methylbutane with cyanide ion? (a) The CN concentration is halved, and the 1-iodo-2-methylbutanc...
-
Leah, age 50, is married with no taxable compensation for 2023. She and her spouse, age 48, reported taxable compensation of $60,000 on their 2023 joint return. Leah may contribute what amount to her...
-
Briefly define the five filing categories available to taxpayers. When might married taxpayers choose to file separately? 2. Distinguish between gross earnings and take-home pay. What does the...
-
How could a typical income statement be usefully visualized as a chart? Explain.

Study smarter with the SolutionInn App


