(a) Write a Lewis structure for HNO 2 in Eq. 23.50. (b) Write a mechanism for the...
Question:
(a) Write a Lewis structure for HNO2 in Eq. 23.50.
(b) Write a mechanism for the reaction shown in Eq. 23.50.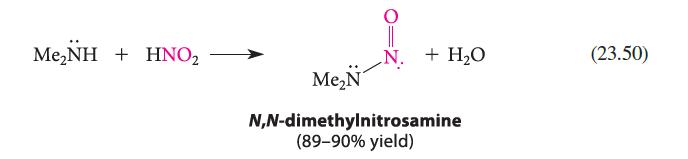
Transcribed Image Text:
Me,NH + HNO, + H₂O Me₂N N,N-dimethylnitrosamine (89-90% yield) (23.50)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a b OH M...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a mechanism for the reaction shown in eq. 10.35. room C-OCH, +HCl (10.35) temp benzoyl chloride methyl benzoate
-
(a) According to the mechanism of the reaction shown in Eq. 10.24, what would be the absolute configuration of the alkyl chloride obtained from the reaction of thionyl chloride. With (S)...
-
Write the Lewis structure for each of the following species, describe its geometry, and indicate the oxidation state of the nitrogen: (a) HNO2 (b) N3- (c) N2H5+ (d) NO3-
-
The following inventory information is available for Ricci Manufacturing Corporation for the year ended December 31, 2017: Beginning Ending Inventories: Raw materials Work in process Finished goods...
-
Name and describe six areas of specialization for a managerial accountant.
-
What conclusions can the auditor make from a negative confirmation?
-
Reconsider Problem 65. Plot a graph of capital recovery cost versus useful life for lives 1 to 25 by 1. Data from problem 65 Crush Autosmashers can purchase a new electromagnet for moving cars at a...
-
The Ambrosia Bakery makes cakes for freezing and subsequent sale. The bakery, which operates five days a week, 52 weeks a year, can produce cakes at the rate of 116 cakes per day. The bakery sets up...
-
__________ are expenses that can be subtracted from total income in order to calculate tax liability. They may include child care expenses and union dues. deductions debentures tax credits dividends...
-
Design a synthesis of methyl orange (Eq. 23.49) using aniline as the only aromatic starting material. -N=N- methyl orange (yellow) an azo dye -NMe H3O+ NHN protonated methyl orange (red) pka = 3.5...
-
As shown in the following equation, when (R)-1-deuterio-1-butanamine is diazotized with nitrous acid in water, the alcohol product formed has the S configuration (D = 2 H). (a) Give the...
-
Hand trace an unordered list through the following operations. X.addToFront(new Integer(4)); X.addToRear(new Integer(7)); Object Y = X.first(); X.addAfter(new Integer(3), new Integer(4));...
-
On June 1st, 2020, Coca Cola (KO) issued bonds for one billion dollars. The bonds have a par value of $1,000, coupon rate of 2.75% payable semiannually, and 40 years maturity (maturity date:...
-
A 9-volt battery is connected to a lightbulb. The lightbulb has a resistance of 50 ohms. What is the current that flows through the lightbulb?
-
What are the qualities of Wendy's verbal and nonverbal communication that can be considered most effective What aspects of the speech (delivery and/or content) could be more effective How does Wendy...
-
3) A violin's E string vibrates at 660 Hz, the distance between clamps is 330 mm, and -4 H = 4x10k. What is the tension at resonance? m 4) What length would the string in Question 3 need to be to...
-
a). Altman Corporation has interest expenses of $120,000 annually. Altmans annual sales are $4 million, its tax rate is 25%, and its net profit margin on sales is 10 percent. What is Altmans TIE? b)....
-
What is the purpose of an approval committee? Who is usually on this committee?
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
When tert-butyl bromide undergoes SN1 hydrolysis, adding a "common ion" (e.g., NaBr) to the aqueous solution has no effect on the rate. On the other hand, when (C6H5)2CHBr undergoes SN1 hydrolysis,...
-
When the alkyl bromides (listed here) were subjected to hydrolysis in a mixture of ethanol and water (80% EtOH/20% H2O) at 55oC, the rates of the reaction showed the following order: (CH3)3CBr >...
-
The reaction of 1o alkyl halides with nitrite salts produces both RNO2 and RONO. Account for this behavior.
-
Background As you have learned during the course, regulatory compliance requires constant vigilance by the board of directors, company executives, and employees. Constant vigilance, in turn, requires...
-
A firm evaluates all of its projects by applying the IRR rule. Year Cash Flow 156,000 0 60,000 1 79,000 2 63,000 3 What is the project's IRR? (Do not round intermediate calculations and enter your...
-
each of the series of questions below based upon the factual scenarios presented above them. Fact Pattern #1 You decide to join with one or more of your fellow classmates to open and operate a...

Study smarter with the SolutionInn App


