Arrange the following three amines in order of increasing basicity. Cl- -NH Cl- -NH p-chloroaniline 4-chlorocyclohexanamine -NH
Question:
Arrange the following three amines in order of increasing basicity.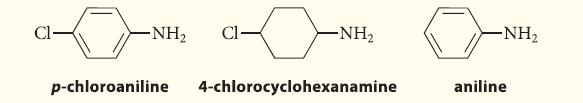
Transcribed Image Text:
Cl- -NH₂ Cl- -NH₂ p-chloroaniline 4-chlorocyclohexanamine -NH₂ aniline
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Because the chloro substituent is an electronwithdrawing group it reduces the basicity of an aniline ...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the amines within each set in order of increasing basicity in aqueous solution, least basic first. (a) Propylamine, ammonia, dipropylamine (b) Methyl 3-aminopropanoate, sec-butylamine, (c)...
-
(a) Arrange the following compounds in order of decreasing acidity and explain your answer: CH3CH2NH2, CH3CH2OH, and CH3CH2CH3. (b) Arrange the conjugate bases of the acids given in part (a) in order...
-
Place the following compounds (a) in order of increasing basicity and (b) in order of increasing acidity. NHCCH3 NH2 NH2 acetanilide cyclohexylamine aniline
-
Strike Bowling installs automatic scorekeeping equipment with an invoice cost of $180,000. The electrical work required for the installation costs $8,000. Additional costs are $3,000 for delivery and...
-
What transactions might be included in a multiple-column purchases journal that would not be included in a single column purchases journal? Discuss.
-
Under what circumstances do attest reports have limited distribution?
-
Quilts R Us (QRU) is considering investing in a new patterning attachment with the cash flow profile shown in the table below. QRU's MARR is 13.5 percent/year. a. What is this investment's external...
-
The bank portion of the bank reconciliation for Rintala Company at November 30, 2014, was as follows. The adjusted cash balance per bank agreed with the cash balance per books at November 30. The...
-
A center that has been in operation for five years needs Group of answer choices only an operating budget for the upcoming year only a long-range financial plan that includes a 10 year projection on...
-
Draw the structure of each of the following compounds. (a) N-isopropylaniline (b) Tert-butylamine (c) 3-methoxypiperidine (d) 2,2-dimethyl-3-hexanamine (e) Ethyl 2-(diethylamino)pentanoate (f)...
-
Clavulanic acid (Fig. P22.98) is an inhibitor of b-lactamase enzymes, which cause penicillin resistance. Clavulanic acid inhibits b-lactamases by first reacting with the side-chain hydroxy group of...
-
a) Why is vulnerability testing desirable? b) What two things does vulnerability testing software do? c) Why is it important to get approval in writing before conducting a vulnerability test? d) What...
-
Define case management . Include who case management serves and the goals of case management.
-
What is the difference between a transfer tax and a business tax? What is the difference between value added tax and a percentage tax? What is the difference between an excise tax and a documentary...
-
A new wine company can lease a storefront or purchase a small building. The lease will cost $1,700 per month and run for ten years. The alternative is to purchase a building by taking out a loan for...
-
What rules govern the internal management of a company? Can these rules be varied? If so, how?
-
Suppose a man who has been accused of a crime cannot afford a lawyer to help with his defense and the officers who have detained him have not allowed him to call anyone; they also have not provided...
-
A major retail store recently spent $24 million dollars on a large private satellite communication system that provides state-of-the-art voice, data, and video transmission between stores and...
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
Compounds L and M have the molecular formula C7H14, Compounds L and M are optically inactive, are nonresolvable, and are diastereomers of each other. Catalytic hydrogenation of either L or M yields...
-
How many stereoisomers are possible for 4-methyl-2-hexene, and how many fractions would you obtain if you distilled the mixture?
-
Propose structures for compounds E-H. Compound E has the molecular formula C5H8 and is optically active. On catalytic hydrogenation E yields F. Compound F has the molecular formula C5H10, is...
-
For questions 8 and 9, consider the graph below for a firm operating in a perfectly competitive market: Price $815 $750. $500 $375 8a.) Calculate the firm's total revenue. 8b.) Calculate the firm's...
-
Z. Ting & Mohammed v. Fearsome White-Water Rafting Ltd. Zhao Ting and Henry Mohammed set out for a day of fun at the Fearsome White-Water Rafting attraction. They both signed a waiver stating they...
-
break this up into 5 slides for a powerpoint presentation.. the order should be logical and engaging. each slide should have bullet points/short sentences. "Introduction The purpose of this report is...

Study smarter with the SolutionInn App


