Complete the reactions given in Fig. P27.75 by giving the structure of the major organic product(s). (a)
Question:
Complete the reactions given in Fig. P27.75 by giving the structure of the major organic product(s).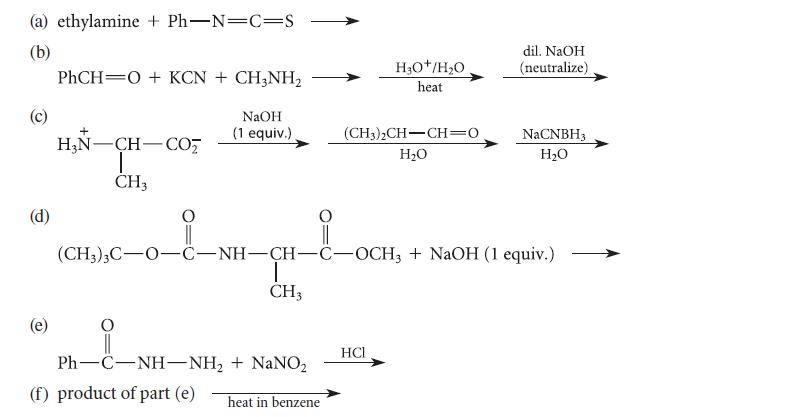
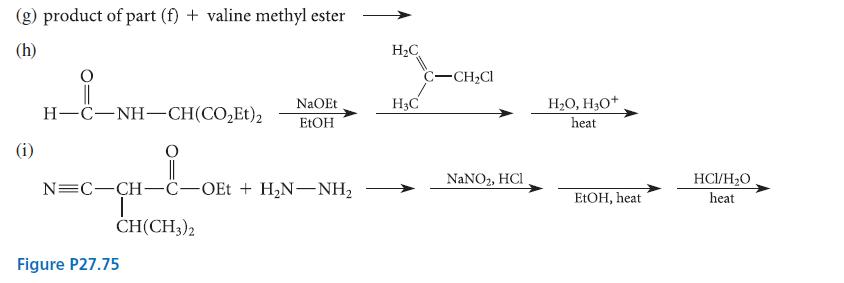
Transcribed Image Text:
(a) ethylamine + Ph-N-C=S (b) (d) PhCH=O + KCN + CH3NH, NaOH (1 equiv.) + HẠN—CH-CO; I CH3 CH3 Ph—C-NH-NH, + NaNO, heat in benzene (f) product of part (e) H₂O+/H₂O heat (CH3)2CH-CH=0 H₂O _0_&_M_H_E_ (CH3)3C-0-C-NH-CH-C-OCH3 + NaOH (1 equiv.) HCI dil. NaOH (neutralize) NaCNBH3 H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a b c d This is like the reaction of an amine with an isocyanate see Eq 2373 on text p 1218 ENHCNHPH This is a modified Strecker synthesis in which me...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete the reactions given in Fig. P22.81 by giving the major organic products. Explain your reasoning. NaOEt excess) EtOH H,o heat CHsI (c) CI NO, H ot, heat C CH LiAIH 2) HaO (e) CH Et CHCHCOEOH...
-
Complete the reactions given in Fig. P19.45 by giving the principal organic product(s). Fig. P19.45 (a) (b) (c) p-toluenesulfonic acid (catalyst CH, t CH,OH - (solvent) ether Hio -caphenone, +...
-
Complete the reactions given in Fig. P21.52 by giving the principal organic products. Explain how you arrived at your answers. NaOH CH O (trace) H,C CCHO CH +CH OH (solvent) Ph NH2 1 (CgH,NO3)...
-
EVE, Inc. reported a return on investment of 20%, a sales margin of 25%, and residual income of $480,000. The required rate of return on invested capital is 8%. On the basis of this information, the...
-
Why is depreciation expense added to net income (indirect method) on the statement of cash flows?
-
Use the following information to answer the questions that follow. a. Use the DuPont system to compare the two heavy metal companies shown above (HMM and MS) during 2012. Which of the two has a...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
Chris Spear invested $15,000 today in a fund that earns 8% compounded annually. To what amount will the investment grow in 3 years? To what amount would the investment grow in 3 years if the fund...
-
3. (25 points) Consider two firms out of a competitive industry. They have the following technologies: C(y) = y + 2y; C2(y) = 1.5y + 3y. Show these firms' individual supply functions on a...
-
Complete the reactions given in Fig. P27.70 assuming the amino acid residue is part of a peptide in aqueous solution and is at neither the amino nor the carboxy terminus. (a) lysine residue + HC=O +...
-
Following is a ribbon diagram for one type of opioid receptor, a family of proteins that bind morphine and other opioids and initiate their physiological effects. The opioid receptor consists mostly...
-
Phillips Company manufactures air-conditioning units for commercial buildings and has noticed considerable variation in shipping expenses from month to month as per the data below: If 10 air...
-
O Macmillan Learning Add the numbers and round the answer to the correct number of significant figures. 112.145+ 45.35 = I
-
! Required information [The following information applies to the questions displayed below.] On January 1, 2024, the general ledger of ACME Fireworks includes the following account balances: Accounts...
-
1. Only the Michigan Legislature, as part of the legislative branch of government, makes laws affecting employment. ?explain 2. Some statutes require a plaintiff to exhaust all administrative...
-
Your client, Ashkan Shirazi, a citizen of Iran, applied under the Federal Skilled Worker Class. Among other facts, the applicant's father lives in St. John's, Newfoundland with his wife. They are...
-
Assume that by June, the price of crude palm oil in the cash market has fallen to RM2,245 per metric tonne and the June crude palm oil futures is also trading at the same price. The producer closes...
-
The Greek Corporation makes two products: Kappa and Gamma. Although each product uses a different type of raw material, the firm produces both products in its Eastern plant. The products make use of...
-
The value of a share of common stock depends on the cash flows it is expected to provide, and those flows consist of the dividends the investor receives each year while holding the stock and the...
-
(a) If the standard enthalpy change for the reaction 2-ethyl- I -butene I -hexene is + I 5.3 kJ mol-r (+3.66 kcal mol-t). and if Afli for l-hexere is -40.5 kJ mol-r (-9.68 kcal mol-1 2-methy...
-
Within each series arrange the compounds in order of increasing stability: C(CH3)3 HO CH,CH(CH)
-
Give the structure for each of the following: (a) 2-methylpropene (b) 5 -(3 -pentenyl)- 1, 3,6,8-decatetraene
-
What are some advantages and disadvantages of a cyber security policy in an HR manual?
-
Create your own radio spot! Include 50 words in your script. At the beginning and the end write down what song you would use to grab attention or you could add a sponsor. You can create a concert...
-
What body system(s) are directly impacted by Type I Diabetes Mellitus and how are those systems affected?

Study smarter with the SolutionInn App


