Consider the following mechanism for Eq. 18.19. Identify the process associated with each step. Counting electrons at
Question:
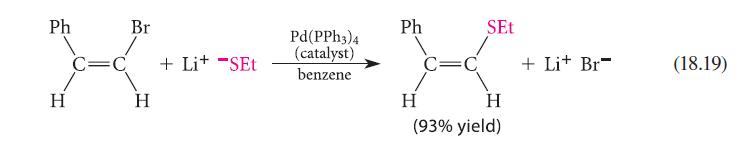
Consider the following mechanism for Eq. 18.19. Identify the process associated with each step. Counting electrons at each stage may help you.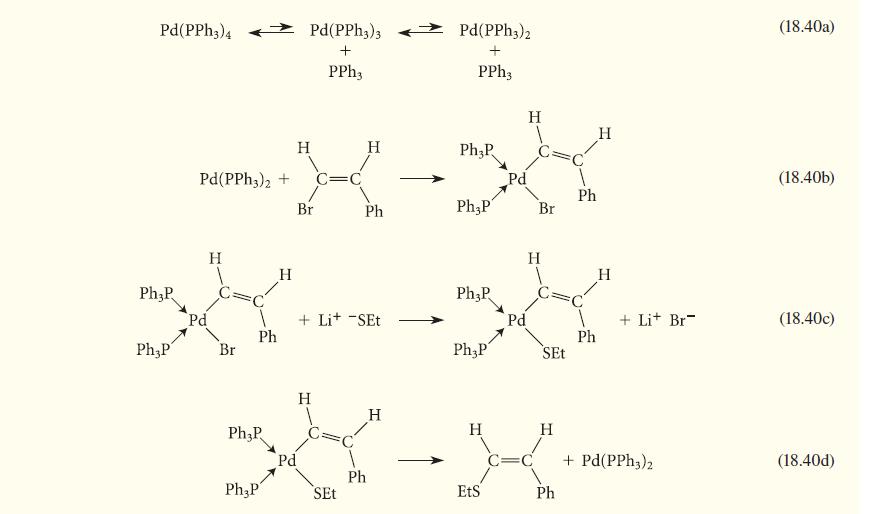
Transcribed Image Text:
Pd(PPH3)4 Ph3P Ph,P Pd(PPh 3)2 + Pd H Br Ph H Ph₂P Pd(PPH3)3 + PPh3 H Br C=C H Ph + Lit -SET Pd(PPH3)2 + PPh3 Ph₂P Ph,P Ph₂P Ph P Pd Pd H Br H SEt XXX-X Ph Ph H H + Lit Br + Pd(PPH3)2 (18.40a) (18.40b) (18.40c) (18.40d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Step 1840a consists of two successive ligand dissociations that reduce the ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the process evaluation article that you chose and explain why you selected this example. Describe the purpose of the evaluation, the informants, the questions asked, and the results of the...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Dickens, Kristen, is enrolled as a doctoral student in the Counselor Education at the University of New Orleans. She is a registered counselor intern in the state of Louisiana and works at a...
-
Implement the method contains() for HashST.
-
1. For the principal quantum number n = 3, what are the possible values of the quantum numbers and m? 2. What is the energy of the shortest wavelength photon emitted by the hydrogen atom?
-
The Cutler Corporation prepared the following balance sheet: Required 1. Identify the errors made in the Cutler balance sheet. 2. Prepare a corrected, properly classified balancesheet. CUTLER...
-
Compute the utilization for these task sets: a. P1: period = 1 s, execution time = 10 ms; P2: period = 100 ms, execution time 10 ms b. P1: period 100 ms, execution time = 25 ms; P2: period = 80 ms,...
-
Bechtel Technical Clothing produces parkas used by arctic explorers, mountain climbers, and people living in Minnesota. The company uses a standard costing system, and standards call for 3.5 yards of...
-
How does the complement system participate in both innate and adaptive immunity, mediating opsonization, inflammation, and the formation of membrane attack complexes?
-
Arrange the following compounds according to increasing rate of elimination with NaOEt in EtOH. What is the product in each case? Ph H C=C A Ph Br Ph H C=C B Br Ph Ph H C=C C Ph Cl H H =C D Br Ph
-
The reaction given in Fig. P17.59 occurs by a mechanism called the S N 2 mechanism, which is a bimolecular substitution that occurs by reaction of the nucleophile at an allylic carbon. In this...
-
Weather Aerodynamic, Inc., makes electronically equipped weather-detecting balloons for university meteorology departments. Because of recent nationwide inflation, the companys management has ordered...
-
You are given the following information on a proposed cigar tax in the Republic of Panatela. Panatela collects no other specific excise taxes, and all other government revenues come from a neutral...
-
Some of the same researchers whose work is described also did another study looking at the outcomes of households that moved versus those that did not in the general population. To control for...
-
Why do you think there are so few firms in this business in the first place?
-
The town of Mt. Pilot has a three-member town council that is considering new infrastructure projects for the town: building a new park, buying a new fire truck, and repaving Main Street. The town...
-
On 2 January 20x1, Alcato Company introduced an executive share option plan for its top ten executives. Under the plan, each executive was granted 100,000 share options that gave them the right to...
-
Shown here are the top 19 companies in the world in terms of oil refining capacity. Some of the companies are privately owned and others are state owned. Suppose six companies are randomly selected....
-
In the circuit shown in Figure 4, a battery supplies a constant voltage of 40 V, the inductance is 2 H, the resistance is 10, and l(0) = 0. (a) Find l(t). (b) Find the current after 0.1s.
-
A chemist finds that the addition of (+)-epinephrine to the catalytic reduction of butan-2-one (Figure 5-16) gives a product that is slightly optically active, with a specific rotation of +0.45o...
-
1. Make a model of each compound, draw it in its most symmetric conformation, and determine whether it is capable of showing optical activity. (a) 1-bromo-1-chloroethane (b) 1-bromo-2-chloroethane...
-
Draw three-dimensional representations of the following compounds. Which have asymmetric carbon atoms? Which have no asymmetric carbons but are chiral anyway? Use your models for parts (a) through...
-
3. For the following array of test scores, [99,58,87,78,100,100,91,93,78,66] Find the mean, standard deviation, minimum value, maximum value, and total number of scores using MATLAB's built-in...
-
First, give a brief of the case. Then explain what are some of the marketing strategies that the company uses to differentiate itself from competitors? What are some of the specific customer...
-
(15 points) Write the following code segment in MARIE's assembly language: if X> 5 then endif; Y=X+X+X; X=0; Y-Y-1;

Study smarter with the SolutionInn App


