Determine whether the following compound, isolated from the frontal gland secretion of a termite soldier, is a
Question:
Determine whether the following compound, isolated from the frontal gland secretion of a termite soldier, is a terpene.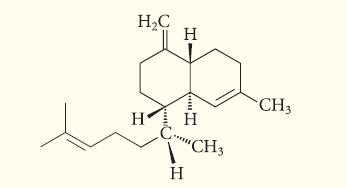
Transcribed Image Text:
H₂C Η Η mm) H Η: Η 'CH, `CH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Because stereochemistry is not an issue delete all stereochemical details for simplici...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether the following symmetric matrices are positive definite by computing their eigenvalues. Validate your conclusions by using the methods from Chapter 4. (a) (b) (c) (d) 3 2 011 121 110...
-
(a) Determine whether the following compound (zoapatanol, used as a fertility-regulating agent in Mexican folk medicine) is a terpene. (b) What product is obtained when zoapatanol is subjected to MnO...
-
1.Why are teenagers more likely to throw temper tantrum? (a.) Because their frontal lobe is not well developed yet b. Because their temporal lobe is not well developed yet c. Because their occipital...
-
What is the coefficient of sliding friction and what is a representative value for this coefficient for the brittle crust?
-
Effect of borrowing and interest on statement of cash flows Gillette borrowed $250 mil lion on October 1 by issuing bonds. The debt carries an annual interest rate of 6%, which it must pay on April 1...
-
Alpha Pro Company is a marketing firm. The company's trial balance on July 31, 2011, appears below. During the month of August, the company completed the following transactions: Aug. 2 Paid rent for...
-
Thrust is the force that pushes a rocket forward. It is a force exerted on the rocket as the engine expels hot gases from the rear of the rocket. For most rockets, thrust is variable rather than...
-
Jordan Enterprises is considering a capital expenditure that requires an initial investment of $42,000 and returns after-tax cash inflows of $7,000 per year for 10 years. The firm has a maximum...
-
For the last section of the project, you will complete the following: Develop a training strategy by including all of the following: Objectives Target audience Delivery method Evaluate the...
-
What products are expected in the reaction of H 2 C = CHCH 2 CH 2 CH 2 CH 3 (1-hexene) with NBS in CCl 4 in the presence of peroxides? Explain your answer.
-
Propose a curved-arrow mechanism for the reaction given in Fig. P16.66. Figure P16.66 (CH3)3C- -C(CH3)3 + HNO3 (CH3)3C- -NO + (CH3)2C=CH
-
Given that find the values of A, B,C and D. 9x3-11x + 8x - 4 x(3x - 2) = B C A++ + x X D 3x - 2
-
Compute MACRS depreciation for the following qualified assets for the calendar years 2019 and 2020: (Ignore bonus depreciation and the Section 179 deduction.) Asset Business equipment (7-year...
-
In 2019, Jason Products Co., a calendar year taxpayer, purchased business equipment (7-year property) for $3,400,000. No other personal property was purchased during the year. Jason wants to take the...
-
Refer to the facts in Problem 40 and assume the property purchased is nonresidential rental property. Compute MACRS depreciation on the property for 2019 and 2020, assuming the property is not...
-
Prepare a brief written abstract for each of the following additional selected court cases relating to allowable trade or business deductions. a. A. Trujillo, 68 TC 670, Dec. 34,554. b. Illinois...
-
The following selected court cases have helped to shape the tax law in regard to allowable trade or business tax deductions. Read the following court cases and prepare a brief written abstract for...
-
For the two large newspapers in your city, you are interested in knowing whether there is a significant difference in the average number of pages in each dedicated solely to advertising. You randomly...
-
Recall that Chapter 8 described the binary search algorithm for finding a particular entry in an ordered list. The idea behind binary search is to begin looking in the exact center of the list. If...
-
The 60-MHz proton NMR spectrum of 2, 2, 3, 3-tetra-chlorobutane consists of a sharp singlet at 25oC, but at -45oC consists of two singlets of different intensities separated by about 10 Hz. Explain...
-
What changes would you expect in the 13C NMR spectrum of 1-bromopropane upon cooling the compound to very low temperature?
-
In the following set, the NMR spectra of the compounds shown consist of a single resonance. Arrange the compounds in order of increasing chemical shift, smallest first. CH,CI, CH212 CH31
-
If engineering management is broadly defined to include the generalmanagement responsibilities engineers can grow into, how it differs from ordinarymanagement? Using an example that you are aware of,...
-
KDS is an engineering company which is organised for managementpurposes in the form of several autonomous divisions. The performance ofeach division is currently measured by calculation of its return...

Study smarter with the SolutionInn App


