Estimate the standard enthalpies of the reactions shown in Eqs. 5.67 and 5.68. (CH3)3C-O + H-Br tert-butoxy
Question:
Estimate the standard enthalpies of the reactions shown in Eqs. 5.67 and 5.68.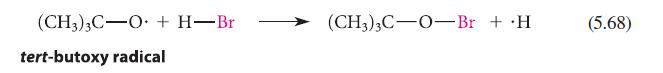
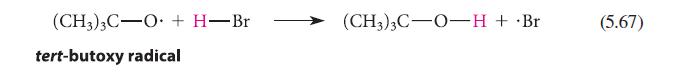
Transcribed Image Text:
(CH3)3C-O + H-Br tert-butoxy radical (CH3)3C-O-Br + .H (5.68)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To obtain the required estimates apply Eq 569 In both equations the bond broken is the HBr bond From Table 53 the bond dissociation energy of this bon...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The standard enthalpies of formation of S(g), F(g), SF4(g), and SF6(g) are 1278.8 kJ/ mol, 179.0 kJ/ mol, 775 kJ/ mol, and 1209 kJ/ mol, respectively. a. Use these data to estimate the energy of an...
-
The standard free energies of formation and the standard enthalpies of formation at 298 K for difluoroacetylene (C2F2) and hexafluorobenzene (C6F6) are For the following reaction: C6F6(g) 3C2F2(g) a....
-
The standard enthalpies of formation of ClO and ClO2 are 101 and 102 kJ/mol, respectively. Using these data and the thermodynamic data in Appendix C, calculate the overall enthalpy change for each...
-
Refer to the table for Moola at the bottom of this page to answer the following questions. What is the equilibrium interest rate in Moola? What is the level of investment at the equilibrium interest...
-
Harrison Ford Company has been approached by a new customer with an offer to purchase 10,000 units of its model IJ4 at a price of $4 each. The new customer is geographically separated from the...
-
During the 1980s and 1990s, changes in the S&P 500 (a broadly diversified index of U.S. stocks) were about 50% correlated with the MSCI EAFE Index (a broadly diversified index of European and other...
-
Assume the same data as given in problem 9, except the company expects the following production: Case A: 300 bbl per month Case B: 500 bbl per month REQUIRED: a. Determine the number of months needed...
-
Lott Company uses a job order cost system and applies overhead to production on the basis of direct labor costs. On January 1, 2017, Job 50 was the only job in process. The costs incurred prior to...
-
An l-regular graph, l 1, is a simple graph where all of its vertices have degree l. The union of two simple graphs G G U G2 = (V U V2, E1 U E2). = (V1, E1) and G2 = (V2, E2), is the simple graph Two...
-
Give the products expected when each of the following alkenes is subjected to oxymercurationreduction. (a) Cyclohexene (b) 2-methyl-2-pentene (c) Trans-4-methyl-2-pentene
-
Alkenes undergo the addition of thiols at high temperature in the presence of peroxides or other free-radical initiators. The following reaction is an example. Propose a mechanism for this reaction....
-
This problem reconstructs the trajectory r 0 (t) of a charged particle from the fields produced by the particle at observation points where the magnetic field does not vanish. (a) Use cB = n ret E...
-
Interactive Investments has one of its new clients, Jason Todd, recently sign up with the firm to manage his portfolio. Jason recently came into an inheritance of $1,250,000 that he wants to have...
-
Calculate discount factors from project A . Project A 10% discount Present value Year Cash flow () factor = cash flow () 0 -1500 1 = -1 500 1 +500 0.9091 = + 454.55 2 +800 0.8264 + 661.12 3 +1 000...
-
List four stakeholders of a company and their objectives. Give one example when the objectives of two stakeholders are not Aligned .
-
Jan buys a $1000 10 percent semi-annual coupon bond for a price of $1094 and holds it to maturity in six years. What is the bond's yield to maturity?
-
Which inventory method is logical for companies to follow to reduce the risk of the inventory becoming out of date?
-
The following schedule relates the income statement with cash flows from operating activities, derived by both the direct and indirect methods, in the format illustrated by Graphic 21-10 in the...
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
For each pair of compounds, explain which is the stronger acid?
-
Explain why the compound on the left is a stronger acid than the compound on the right.
-
Provide IUPAC name for these alkenes: CH3 a) CHCHCHCHCH CH3 c) CHCHCHCHCH3 e) CHCH3 CH3 b) CHCHCHCHCH CHCH3 d) CHCHCHCHCCHCHCHCH T CH, CHCH CH, f)
-
R , A coin with a center hole is wobbling about a frictionless pole. A torque T is applied to the coin and is always along the central axis (blue) of the coin. At the contact point p between the coin...
-
Fawn Hollow LLC, is owned equally by three individuals, Mark Crenshaw, Thomas Miller, and Jerald Wolfowicz. The LLC was formed in 2014 for the purpose of building and operating a hotel property with...
-
p(z) = p(0) e (z/H) 1) (20 PTS) Using the equation above, show that the total mass m of the species in the atmosphere is given by: m = A H. p(0) where: A surface area of the Earth (Re = 6371 km). 2)...

Study smarter with the SolutionInn App


