For each of the following balanced oxidationreduction reactions, indicate which compound(s) are oxidized and which are reduced.
Question:
For each of the following balanced oxidation–reduction reactions, indicate which compound(s) are oxidized and which are reduced.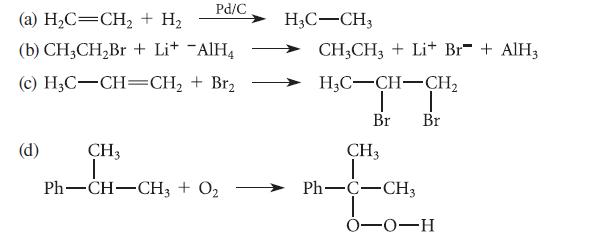
Transcribed Image Text:
Pd/C (a) H₂C=CH₂ + H₂ (b) CH3CH₂Br + Li+ AlH4 (c) H3C-CH=CH₂ + Br₂ (d) CH3 T Ph–CH–CH3 + O2 H3C-CH3 CH3CH3 +Li+ Br+AlH3 H3C-CH-CH₂ I T Br Br CH3 Ph—C–CH3 0-0-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a The alkene is reduced and the H is oxidi...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
KitchenPaid (KP) is a U.S. manufacturer of upscale small kitchen appliances. As a result of an acquisition made in 2013 KP also has a small division that manufactures medical supplies. KP is...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) (b) (c) (d) ...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) CH3C = PCH +...
-
Doug, Peter, and Jack have the following capital balances;$150,000, $300,000 and $320,000, respectively. The partners shareprofits and losses 35%, 40%, and 25% respectively. Jones is goingto pay a 2...
-
Refer to the data in PE 6-16. Calculate the companys average collection period. Data from PE 6-16 Accounts receivable balance, December 31 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
The notion of a market for information, unlike markets for agricultural commodities, transportation services, and so on, may be unfamiliar to most people. A main reason for this is that information...
-
Tyler Company reported the following costs on its financial statements (in thousands): REQUIRED: Using the reserve disclosure for Tyler Company in problem 13 and the data presented in this problem,...
-
Your division is considering two projects. Its WACC is 10%, and the projects? after-tax cash flows (in millions of dollars) would be as follows: a. Calculate the projects? NPVs, IRRs, MIRRs, regular...
-
Analyses at least two considerations that would be required to ensure effective transaction transparency of centralized database management systems and distributed database management systems....
-
Contrast the products expected when 2-methyl-3-pentanol is treated with (a) HBr/H 2 SO 4 or (b) Ph 3 PBr 2 . Explain.
-
Give the structure of two secondary alcohols that could be converted by HBr/H 2 SO 4 into the corresponding alkyl bromide without rearrangement.
-
A simple random sample of size n is to be taken without replacement from a population of size N. a. Determine the probability that any particular sample of size n is the one selected. b. Determine...
-
Assume that Freezeqwik Ltd (Activity 10.16) wishes to reduce its OCC by 30 days. Evaluate each of the options available to this business.
-
The following figures relate to the retail business of Daisy King for the month of July. Goods sold fall into two categories, X and Y. You are to calculate for each category of goods: (a) Cost of...
-
Styrene can be hydrogenated to ethyl benzene at moderate conditions in both the liquid and the gas phases. Calculate the equilibrium compositions in the vapor and liquid phases of hydrogen, styrene,...
-
A home heating system maintains the indoor temperature at 20 C. The outside temperature is 0 C. A furnace burns natural gas to heat a 1:4 mixture of outside and inside air. The resulting air is at a...
-
Yaso is in business buying and selling goods on credit. He is concerned that although his business is making a good profit, his balance at the bank is not increasing. The following information is...
-
Adrian is a salesperson who represents several wholesale companies. On January 2, 2011, she receives by mail a commission check from Ace Distributors in the amount of $10,000 and dated December 30,...
-
Identify the tax issues or problems suggested by the following situations. State each issue as a question. Jennifer did not file a tax return for 2007 because she honestly believed that no tax was...
-
Write the equation for the final step in the -oxidation pathway of any fatty acid with an even number of carbon atoms.
-
Show the products of each of the followingreactions: (a) FAD FADH2 CHCH-CH2CH2CHzCsCOA Acyl-CoA dehydrogenase (b) Enoyl-CoA hydratase Product of (a) + H20 NAD+ NADH/H* (c) Product of (b)...
-
What is the structure of the a-keto acid formed by transamination of each of the following amino acids? (a) Threonine (b) Phenylalanine (c) Asparagine
-
(a) Show without using 25in a calculator that: = Sin -455 9 (b) 2 cos" (+1) = cos `"` (-3)
-
A furniture company is considering investing in several projects to expand its business. The projects under consideration are (1) Lights and/or (2) Shelving. Both projects require an initial...
-
195 Es) Suppose that the algebraic expression for the z-transform of xn = 1-2 2 (1 + ) (1 + + ) nt regions of convergence.co 2

Study smarter with the SolutionInn App


