For the reactions given in Fig. P18.86, explain why different products are obtained when different amounts of
Question:
For the reactions given in Fig. P18.86, explain why different products are obtained when different amounts of AlBr3 catalyst are used.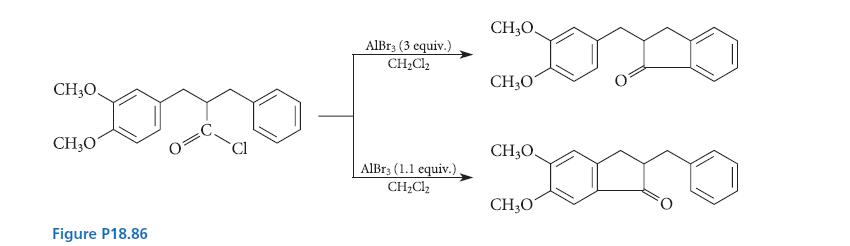
Transcribed Image Text:
CH,O. CH₂O Figure P18.86 AlBrs (3 equiv.) CH₂Cl₂ AlBr3 (1.1 equiv.) CH₂Cl₂ CH,O, Domo CH307 CH3O CH3O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The two reactions are intramolecular FriedelCrafts acylations The first equivalent of AlBr 3 a stron...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Briefly explain why different products are obtained from the electrolysis of molten NaCl and the electrolysis of a dilute aqueous solution of NaCl.
-
Explain why, in the reactions given in Fig. P18.68, p. 883, different stereoisomers of the starting material give different products. Fig. P18.68 oc., (only elimination product observed) H,C Br KOH...
-
The following compound was isolated from nerve cells. (a) Describe how this compound differs in structure from fats and oils. (b) Three products are obtained when this compound is hydrolyzed with...
-
The equity profolio of a company is thus 31/12/2001. Nature: Actions A, Quantity = 350, Unit cost = 36000, Purchase dates = 03/1999, 2000 = 37000, 2001= 34000. Nature: Actions B, Qauntity = 125, Unit...
-
The accounting records of Maura Grayson Architect, P.C., include the following selected, unadjusted balances at March 31: Accounts receivable, $1,400; Supplies, $1,100; Salary payable, $0; Unearned...
-
Entrance to a prestigious MBA program in India is determined by a national test where only the top 10% of the examinees are admitted to the program. Suppose it is known that the scores on this test...
-
Consider the IRR and ERR measures of worth. If we define a root to mean a value for the measure that results in \(\mathrm{PW}=0\), then which of the following statements is true? a. Both IRR and ERR...
-
Qwik Repairs has over 200 auto-maintenance service outlets nationwide. It provides primarily two lines of service: oil changes and brake repair. Oil changerelated services represent 70% of its sales...
-
What ways can I make my Relational Data Model conversion meet the following requirements listed below? Am I on the right track? What corrections do I need to address? ERD: My Conversion of ERD onto a...
-
Give a mechanism for the reaction in Fig. P18.87 by showing the catalytic intermediates. OR OEC CH CH3 Figure P18.87 CH Grubbs Gl catalyst OR + CH3CH=CH
-
Propose a structure for the product A obtained in the following oxidation of 2,4,6-trimethylphenol. (Compound A is an example of a rather unstable type of compound called generally a quinone...
-
Legal action taken by an organization can affect the probability of whether fraud will reoccur. (True/False)
-
How does job analysis support human resource management activities? Briefly describe one of the methods commonly used for gathering job analysis data.
-
You are the manager of the human resources (HR) department. The vice president of finance is talking about an operations issue and then makes a small remark to you about his staff being late or...
-
As a Human Resource Manager for Bank of America in Phoenix, Arizona shortly. The organization (B.O.A) has asked you to evaluate whether the current pay structure makes sense given what competing...
-
You are the VP of Human Resources for an energy company based in Philadelphia, PA. It has about 2,300 employees in a variety of jobs and a union (the IBEW) representing the call center and field...
-
1. Why is it critical to list both systems of measurements on all dasign documentation? 2. Why do quantities in the BUM need to be in the units in which the item is purchased? 3. Why do incoming...
-
Troy Electronics Company calculates its predetermined overhead rate on a quarterly basis. The following estimates were made for the current year. The firms main product, part number A200, requires...
-
Do public and private companies follow the same set of accounting rules? Explain.
-
Condensations in which the active hydrogen compound is a β-keto ester or a β-diketone often yield products that result from one molecule of aldehyde or ketone and two...
-
The following reaction illustrates the Robinson annulation reaction (Section 19.7A). Provide a mechanism. base
-
What is the structure of the cyclic compound that forms after the Michael addition of 1 to 2 in the presence of sodium ethoxide? OEt NaOEt EtO 2
-
On your worksheet, fill in the values of A through N. A through F are denoted by red letters on the previous page and G through N are denoted by red letters on the flowchart below. What does this...
-
The tapered rod has a radius of r as shown below. Determine the average normal stress at the center of the rod: point B. = w (60+40x) lb/in. 3 in. = (2 ) in. T= B -3 in.- x
-
3. Consider a shaft mounted with frictionless bearings at its ends. For T = 400 Nm, TB =175 Nm, and Tc = 225 Nm, the maximum internal torque seen by the shaft is best given by T400 a. 800 Nm b. 225...

Study smarter with the SolutionInn App


