From what you learned in Sec. 1.3B about the relative lengths of CC and CO bonds, predict
Question:
From what you learned in Sec. 1.3B about the relative lengths of C¬C and C¬O bonds, predict which of the following compounds should have the larger energy difference between gauche and anti conformations about the indicated bond. Explain.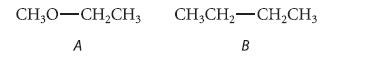
Transcribed Image Text:
CH3O-CH₂CH3 CHỊCH,—CH,CH3 A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The CO bond is somewhat shorter than the CC bond text p 15 ...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Do HF/6-31G* partial geometry optimizations of n-butane conformations with CCCC dihedral angles fixed at several values. Plot the energy versus dihedral angle. From the plot, estimate the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Which one of the following compounds should have a * UV absorption at the greater max when the compound is dissolved in NaOH solution? Explain.
-
The task in this design project is to design an actuation system to power the ram's reciprocating motion in a small-size shaper. The power source of the actuation system is an AC motor with 0.75 hp...
-
How can a judge decide whether there was sexual harassment without holding a trial? We dont know whether Clinton did these things or not.
-
Smith Co. is a firm specializing in financial advice for retired individuals. After some analysis, you have determined that an earnings multiplier of 7 is appropriate for this type of business....
-
Chicago Prime Packers Inc. was a US corporation with its principal place of business in Colorado. Northam Food Trading Company was a Canadian corporation with its principal place of business in...
-
1. Review the financial statements of Victor and Maria Hernandez (Table) and respond to the following questions: (a) Using the data in the Hernandezes balance sheet, calculate an investment...
-
Solve the following complex number problems. Solutions for complex number transformation is required in this item. a. Simplify: 120 +31 18-2115 2111 +5 b. (32-120)+(1/120) (4-1)(-1-1) 5e C. 2e0.25mi...
-
(a) What value is expected for the dipole moment of the anti conformation of 1, 2-dibromoethane, BrCH 2 CH 2 Br? Explain. (b) The dipole moment of any compound that undergoes internal rotation can...
-
(a) Draw Newman projections of the most stable conformations about each of the carboncarbon bonds in the principal chain of 2,2-dimethylpentane. Use models! (b) Combine these to predict the most...
-
How should the joint operators share of assets in the joint operation be disclosed?
-
Explain the Model-View-Controller (MVC) architecture and its implementation in Python web frameworks.
-
In the space below draft an email to Asha Fern putting forward and discussing both tenants, and, using THREE ( 3 ) valid reasons, explain why you feel Sarah and Justine would be the appropriate...
-
AI is a substitute for physical capital for many industries, for example, in the tech industry. Suppose you are Elon Musk and you have to decide how many economists and computer science engineers to...
-
Beginning finished goods inventory $ 7,800 Ending finished goods inventory $ 14,000 Underapplied overhead $ 3,000 Adjusted cost of goods sold $ 53,000 What is the cost of goods manufactured?
-
Sexual harassment continues to be a prevalent problem in a variety of work organizations in Malaysia today, one that has damaging consequences for both individuals and the organizations concerned....
-
For each of the following changes, state what will happen to demand and market equilibrium price and quantity in the short run? a) The price of a substitute good rises. b) Consumer incomes fall, and...
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
Propose structures for molecules that meet the following descriptions. Assume that the kinds of carbons (1, 2, 3, or 4) have been assigned by DEPT-NMR. (a) C 6 H 12 O; IR: 1715 cm 1 ; 13 C NMR: 8.0 ...
-
Compound A, C 8 H 10 O 2 , has an intense JR absorption at 1750 cm ?1 and gives the 13 C NMR spectrum shown. Propose a structure for A. 219 8 TMS 200 180 160 140 120 40 O ppm 80 60 20 100 Chemical...
-
Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 4 H 7 C1O ??IR: 1715 cm ?1 ? (b) C 7 H 14 O ? ? ?IR: 1710 cm ?1 ? (c) C 9 H 10 O 2 ? ? ?IR: 1695 cm ?1 ?...
-
The population of a herd of deer is represented by the function A (t) = 190(1.21)t, where t is given in years. To the nearest whole number, what will the herd population be after 4 years? The herd...
-
Explain how to graph this relation P (r) = 13+2r 2
-
Compute the given integral by first identifying the integral as the volume of a solid. 49 - x - y2 dA, where D is the disk x + y < 49.

Study smarter with the SolutionInn App


