Give the predominant product of the E2 reaction when each of the following diastereomers is allowed to
Question:
Give the predominant product of the E2 reaction when each of the following diastereomers is allowed to react with potassium tert-butoxide in DMSO. Explain your reasoning.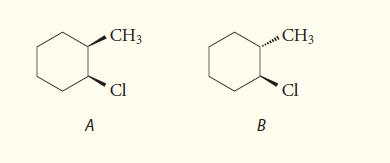
Transcribed Image Text:
A CH3 Cl B CH3 Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Two principles are important in solving this problem The first is that the stereochemistry of the E2 ...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
Most Companys close their books at the end of every calendar month; publicly-listed firms close their books not only every month but every quarter due to the SEC Requirement of the filing of a Form...
-
When ammonia and oxygen are reacted, they produce nitric oxide and water. When 8.5 g of ammonia is allowed to react with an excess of O2, the reaction produces 12.0 g of nitrogen monoxide. What is...
-
Let a = (4, 5) and 6 = (-4,-2). Find the projection of b onto a. proj; b = =V
-
Accounting and financial reporting for state and local governments use, in different places, either the economic resources measurement focus and the accrual basis of accounting or the current...
-
As noted in the chapter, from 1949 to 1990, the Japanese market rose 25,000%. a. Given these returns, does it make sense for Japanese investors to diversify internationally? b. What arguments would...
-
Describe the six categories of early warning signs of potential fraud.
-
During the month of May 20--, The General's Favorite Fishing Hole engaged in the following transactions. These transactions required an expansion of the chart of accounts as shown below. May 1 In...
-
An imaginary Canadian cell phone company would want to examine to determine if there is potential to sell their products in India and USA? How the marketing will be done internationally?
-
The crown ether [18]-crown-6 has a strong affinity for the methylammonium ion, CH 3 + NH 3 . Propose a structure for the complex between [18]-crown-6 and this ion. Show the important interactions...
-
For which of the following alkenes would oxymercurationreduction give (a) A single compound; (b) Two diastereomers; (c) More than one constitutional isomer? Explain. A B -CH3 CH3 CH3 HC a H" D
-
Decide whether each function defined as follows is a probability density function for the given interval. (x) = 0.4; [4, 6.5]
-
What is the difference between an angel investor (angel) and a venture capitalist (VC)?
-
A bicycle manufacturer currently produces 298,000 units a year and expects output levels to remain steady in the future. It buys chains from an outside supplier at a price of $1.90 a chain. The plant...
-
Arnold Inc. is considering a proposal to manufacture high-end protein bars used as food supplements by body builders. The project requires use of an existing warehouse, which the firm acquired three...
-
What are the sources of short-term funding available to large corporations? Differentiate these sources from those needed to raise funds for a long-term capital investment.
-
Southern Textile Ltd. is a manufacturer of sportswear that supplies to a number of European retail stores. Its registered office is in Singapore. During 2017, the firm earned SGD 112,000 before...
-
Lets be more practical. DCF is not the only gospel. Many managers have become too absorbed with DCF. Can such a statement be justified? Why?
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
What product would you obtain from a base-catalyzed Michael reaction of 2, 4-pentanedione with each of the following , -unsaturated acceptors? (a) 2-Cyclohexenone (b) Propene nitrile (c) Ethyl...
-
What Product would you obtain from a base-catalyzed Michael reaction of 3-button-2-one with each of the following nucleophilicdonors? (b) o (a) ELOCH,OET .Et
-
How would you prepare the following compound using a Michaelreaction?
-
In this animated text presentation, the concepts of personal budgeting are outlined. While this is sponsored by a particular financial group, it offers some practical information about this topic....
-
Gary and Brenda both have similar businesses in the garment industry making caps and backpacks. In one day, Gary can make 60 caps and 12 backpacks when he divides his production resources equally...
-
a. Define what is Keynesian Economics and what is Neoliberalism. What is their main difference regarding the role of government in the market place/the economy and why it matters? b. Explain the...

Study smarter with the SolutionInn App


