Give the principal organic product(s) formed in each of the following reactions. (a) o-cresol + Br in
Question:
Give the principal organic product(s) formed in each of the following reactions.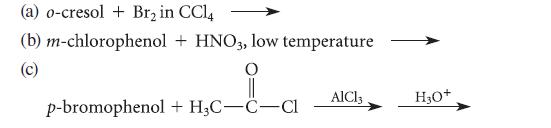
Transcribed Image Text:
(a) o-cresol + Br₂ in CCl4 (b) m-chlorophenol (c) + HNO3, low temperature || p-bromophenol + H₂C-C-Cl AlCl3 H₂O+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a OH Br ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of the principal organic product of each of the following reactions: 2. H NaOH H-O 2. H 3. heat NaOCH.CH.ethanc 1. NaOH, H-O 2. H 3. heat NaOCH CHs. ethanol L. NaOH, HO 2. H 3....
-
Give the structure of the principal organic product of each of the following reactions: (a) (b) (c) (d) (e) COOCH2CH CH,CH2 heat COOCH CH COOCH,CH COOCH,CH NaOCH CH 2. H COOCH2CH3 Product of part (b)...
-
Give the structure of the principal organic product formed on reaction of benzyl bromide with each of the following reagents: (a) Sodium ethoxide (d) Sodium hydrogen sulfide (b) Potassium...
-
Explain how the structure of the balance sheet corresponds to the accounting equation.
-
A student on earth hears a tune on her radio that seems to be coming from a record that is being played too fast. She has a 33-rev/min record of that tune and determines that the tune sounds the same...
-
Tim Smith is shopping for a used car. He has found one priced at $4,500. The dealer has told Tim that if he can come up with a down payment of $500, the dealer will finance the balance of the price...
-
How can poor specifications lead to poor quality codeddo aspects of a poorly constructed specification necessarily lead to bad software?
-
J.P. Morgan Asset Management publishes information about financial investments. Over the past 10 years, the expected return for the S&P 500 was 5.04% with a standard deviation of 19.45% and the...
-
What is the primary reason that auditors' assess internal control?
-
(a) Give the structure of the product formed in the reaction of urushiol with K 2 CO 3 and a large excess of methyl iodide. (b) Would this compound be likely to provoke the same allergic skin...
-
Electron transport takes place in the membrane of cellular organelles called mitochondria. What is it about the structure of ubiquinone and its reduction products that ensures their localization...
-
Sketch the mechanisms of water-soluble and lipid-soluble hormone function.
-
Recently, policymakers have debated whether an increase in the federal minimum wage (currently $7.25 an hour) would be good for the economy. Use a Phillips curve to explain how inflation would change...
-
Using a graphical representation of the new Keynesian model, describe the effects of an unanticipated negative demand shock (label this equilibrium as point 2). Compare these effects to those of an...
-
Illustrate how each of the following will affect aggregate supply and explain your reasoning. a. The implementation of artificial intelligence in manufacturing has led to faster-than-expected...
-
Go to the St. Louis Federal Reserve FRED database, and find data on recession dating (USRECQ) and real GDP (GDPC1), real consumption (PCECC96), and real private domestic investment (GPDIC1). a) Using...
-
Go to the St. Louis Federal Reserve FRED database, and find data on real GDP (GDPC1) and the GDP deflator (GDPDEF). Convert the deflator to the inflation rate by setting the Units setting to Percent...
-
The Public Citizen's Health Research Group studied the serious disciplinary actions that were taken during a recent year on nonfederal medical doctors in the United States. The national average was...
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
Which of the following cycloalkanes are capable of geometric (cis-trans) isomerism? Draw the cis and trans isomers. (a) 3-ethyl-1,1-dimethylcyclohexane (b) 1-ethyl-3-methylcycloheptane (c)...
-
Give IUPAC names for the following cycloalkanes. (a) (b) (c) CH CH,CH,CH, C(CH) CH,CH, CH H C
-
The heat of combustion of cis-1, 2-dimethylcyclopropane is larger than that of the trans isomer. Which isomer is more stable? Use drawings to explain this difference in stability.
-
CEO's horizontal analysis For Short Case #7, CEO has performed horizontal analysis on financials The company financials have shown that sales have been increasing at 3% rate per year, and inventory...
-
Explain that How the concept of organizational silence manifest in different types of organizations?
-
Negative Message Worksheet Instructions Prepare a bad news response based on the scenario. Buffer Positive frame of mind; prepare for transition to bad news Reasons The explanation / justification;...

Study smarter with the SolutionInn App


