Give the product formed when each of the following alcohols is oxidized by dilute HNO 3 .
Question:
Give the product formed when each of the following alcohols is oxidized by dilute HNO3.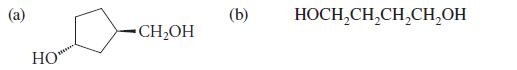
Transcribed Image Text:
HO CH₂OH (b) HOCH₂CH₂CH₂CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
As is the case with carbohydrates ...View the full answer

Answered By

Robert Mwendwa Nzinga
I am a professional accountant with diverse skills in different fields. I am a great academic writer and article writer. I also possess skills in website development and app development. I have over the years amassed skills in project writing, business planning, human resource administration and tutoring in all business related courses.
4.90+
187+ Reviews
378+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the product formed when the following alcohols is oxidized by dilute HNO3. HOCH2CH 2CH2OH
-
Give the product formed when each of the following compounds undergoes an electrocyclic reaction a. Under thermal conditions b. Under photochemical conditions 1. 2. CH3 CH ,.
-
Give the major product formed when each of the following alcohols is heated in the presence of H2SO4; a. b. c. d. e. f. CH3CH2CH2CH2CH2OH CH CH3CH2C CHCH OH CH3 CHCH2CH3 OH CH,OH CH3 CH,CH2CH CCH OH...
-
We can measure how good a center Kevin Bacon is by computing each performer's Hollywood number or average path length. The Hollywood number of Kevin Bacon is the average Bacon number of all the...
-
A particle of rest mass M 0 decays into two identical particles of rest mass m 0 , where m 0 = 0.3M 0 . Prior to the decay, the particle of rest mass M 0 has an energy of 4M 0 c 2 in the laboratory....
-
Reston, Inc., has asked your corporation, Pruro, Inc., for financial assistance. As a long-time customer of Reston, your firm has decided to give that assistance. The question you are debating is...
-
Let \(R\) be a \(\mathrm{BES}^{3}\) process starting from 1. Compute \(\mathbb{E}\left(R_{t}^{-1} ight)\). From the absolute continuity relationship \[\mathbb{E}\left(R_{t}^{-1}...
-
Oregon Products Inc. completed and transferred 72,000 particle board units of production from the Pressing Department. There was no beginning inventory in process in the department. The ending in-...
-
What is the current gdp of India in 2023? difference between 2021-2023. please share all details in index form
-
Give Fischer projections for the aldaric acids derived from both D-glucose and L- gulose. What is the relationship between these structures?
-
An aldohexose A is either d-idose or d-gulose (see Fig. 24.3). It is found that a different aldohexose, L -(2)-glucose, gives the same aldaric acid as A. What is the identity of A? - - H CH=0 -OH HO...
-
Next, for the assertion identified in Question 2, identify a specific internal control activity that would help detect a misstatement related to the recognition of revenue for IRU swaps.
-
Provide an example of how your own consumption would change as the real interest rate changes. Specifically, describe how your opportunity costs change. If other consumers made similar changes, how...
-
On 1 January 20x5, Company A acquired all the 800,000 ordinary shares in Company B for $900,000 in cash. The carrying amount or book value of net assets of Company B as at the date of acquisition...
-
The Fed has conducted expansionary monetary policy to combat a recession but is running up against the zero lower bound, and the economy is still not recovering. What other steps could the government...
-
In 2017, potential output was $18.17 trillion and output was $18.05 trillion. In 2018, potential output was $18.51 trillion and output was $18.56 trillion. Calculate the output gap for each year. How...
-
In 2022, the federal government spent $5.9 trillion and brought in revenue of $4.4 trillion. a. Is this a budget deficit or surplus, and what is its size? b. If net government debt in 2022 was $24.5...
-
An entrepreneur opened a small hardware store in a strip mall. During the first few weeks, business was slow, with the store averaging only one customer every 20 minutes in the morning. Assume that...
-
Simplify the expression. Assume that all variables are positive. 23VI1 2 V44 8
-
Suppose a mixture of AQC-amino acids is subjected to HPLC on a stationary phase that consists of C8-silica rather than Cl 8-silica; that is, the glass stationary phase (Eq. 26.34, p. 1293) contains...
-
Sometimes it is necessary in solid-phase peptide synthesis to use a resin linker that is more sensitive (that is, more reactive) to acid than the linker shown in Eq. 26.21 on p. 1285. The following...
-
Draw the structure of the major neutral form of each of the following peptides, G-D-G-L-F
-
How do you decide upon your timeline? What are the key milestones? How do you ensure your timeline is done in a SMART (specific, measurable, attainable, relevant, and timebound) way?
-
With clear examples, compare and Contrast Constitutional Law and Administrative Law Discuss with case laws that defines the executive power?
-
Determine whether the following statements are true or false. Justify your answers. (a) The set Z7 = {0,1,2,3,4,5,6} is a group under multiplication modulo 7. (b) (c) The relation R on Z defined by...

Study smarter with the SolutionInn App


