Give the products expected (if any) when ethylbenzene reacts under the following conditions. (a) Br in CCl4
Question:
Give the products expected (if any) when ethylbenzene reacts under the following conditions.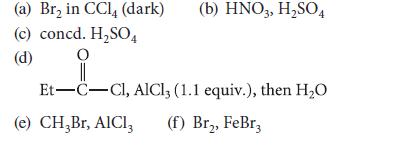
Transcribed Image Text:
(a) Br₂ in CCl4 (dark) (c) concd. H₂SO4 O (d) (b) HNO3, H₂SO4 Et-C-Cl, AlCl3 (1.1 equiv.), then H₂O (e) CH₂Br, AlCl3 (f) Br₂, FeBr3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
In part e ethylbenzene must be present in large excess for the monosubstitution produ...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the products expected when the following compounds are ozonized and reduced. (a) (b) (c) (d)
-
(a) Give the products expected when acetic formic anhydride reacts with (i) aniline and (ii) benzyl alcohol. (b) Propose mechanisms for these reactions.
-
(a) Give the products expected when (+)-glyceraldehyde reacts with HCN. (b) What is the relationship between the products? How might they be separated? (c) Are the products optically active? Explain.
-
In the Akerlof example, the individuals are treated as indifferent to risk. What would you expect to see in these markets if individuals wanted to avoid risk? What if there were some risk lovers?
-
Classification and interpr4eting income statements SeaBreeze Inc., a Taiwan-based semiconductor manufacturer, reported the following information for 2008. SeaBreeze Inc. applies IFRS and reports in...
-
Nella Corporation, which computes depreciation to the nearest whole month, placed a new piece of equipment in operation on July 1, Year 1. It was expected to produce 400,000 units of product in its...
-
You push your \(0.70-\mathrm{kg}\) pillow across your bed with a constant force of \(10 \mathrm{~N}\). The bed provides a frictional force of \(6.0 \mathrm{~N}\). What is the acceleration of the...
-
While completing undergraduate school work in information systems, Dallin Bourne and Michael Banks decided to start a business called ISys Answers which was a technology support company. During year...
-
Describe a situation in which you experienced two decision-making pitfalls. What techniques could be applied in each situation for more effective decision-making? Which decision-making techniques or...
-
Which of the following compounds cannot contain a benzene ring? How do you know? C10H16 A C8H6Cl B C5H4 C C10H15N D
-
Using benzene and any other reagents, outline a synthesis of each of the following compounds. (a) Cyclohexylcyclohexane (b) Tert-butylcyclohexane
-
The following fractions and mixed numbers have repeating decimal equivalent forms. Express their decimal and percent equivalent forms in the repeating decimal notation. Show just the minimum number...
-
Which of the following contracts is riskiest for a buyer? a. time and material b. cost reimbursable c. firm-fixed-price d. fixed-price-economic-price-adjustment
-
Name three different ways to categorize project risks.
-
What does a SWOT analysis examine?
-
One output of the Plan Procurement Management process is the _________________, a document that describes the item to be procured in sufficient detail to allow prospective sellers to determine if...
-
A _________________ analysis is a technique that results in a decision about whether particular work can best be accomplished by the project team or should be purchased from external sources. a....
-
Use the following data to construct a 99% confidence interval to estimate p1 p2. Sample 1 Sample 2 ni-409 P1=.71 n, ,- 378 .67
-
Calculate I, , and a for a 0.0175 m solution of Na 3 PO 4 at 298 K. Assume complete dissociation. How confident are you that your calculated results will agree with experimental results?
-
The following natural product readily gives a Diels-Alder adduct with maleic anhydride (structure in Eq. 15.12a, p. 698) under mild conditions. What is the most likely configuration of the two double...
-
Explain why 4-methyl-1, 3-pentadiene is much less reactive as a diene in Diels-Alder reactions than (E)-1, 3-pentadiene, but its reactivity is similar to that of (Z)-1, 3-pentadiene.
-
This problem describes the result that established the intrinsic preference for L, Z-addition in the reaction of hydrogen halides with conjugated dienes. (a) What is the relationship between the...
-
Internal surface of wall is at 900C Temperature. Wall is 60cm thick, 1m length and 1.5 m width. Thermal conductivity of wall is 0.4 Watt/m-K. Find the thermal resistance and heat transfer rate of...
-
A truck is carrying a refrigerator as shown in the figure. The height of the refrigerator is 158.0 cm, the width is 60.0 cm. The center of gravity of the refrigerator is 69.0 cm above the midpoint of...
-
A deuteron (the nucleus of an isotope of hydrogen) has a mass of 3.3410-27 kg and a charge of 1.6010-19 C. The deuteron travels in a circular path with a radius of 6.50 mm in a magnetic field with a...

Study smarter with the SolutionInn App


