Give the structure of an isomer of the allylic halide reactant in Eq. 17.7 that would react
Question:
Give the structure of an isomer of the allylic halide reactant in Eq. 17.7 that would react with water in an SN1 solvolysis reaction to give the same two products. Explain your reasoning.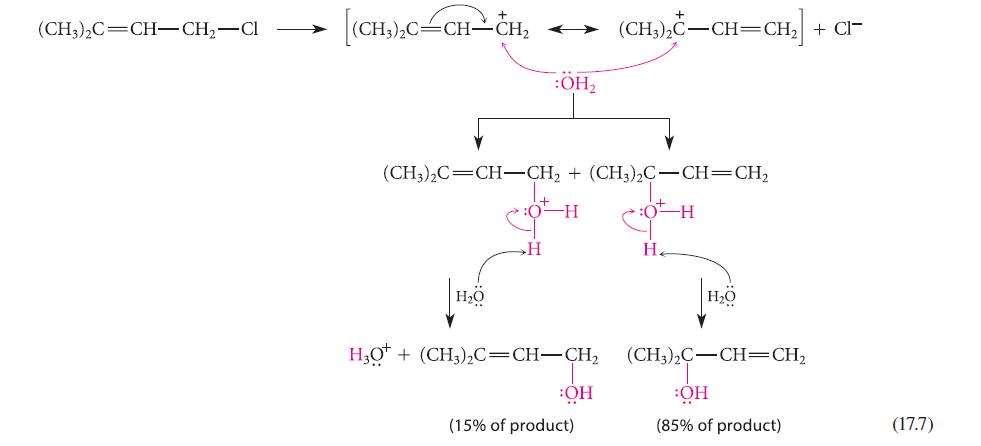
Transcribed Image Text:
(CH3)2C=CH-CH₂-Cl (CH,),C=CH–CH, H₂O :OH₂ (CH3)2C=CH-CH₂ + (CH3)2C-CH=CH₂ :0 H 0-H H HO* + (CH,),C=CH–CH, OH (15% of product) + (CH3)2C-CH=CH₂ + CI- H₂ H₂O (CH;),C–CH=CH, OH (85% of product) (17.7)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The alkyl halide is the one that reacts to give ...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
51+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The S N 1 solvolysis of cinnamyl chloride in water gives two structurally isomeric alcohols (neglect stereoisomers). (a) Show five resonance structures of the carbocation intermediate. In each of...
-
Give the structure of a hydrocarbon that has six carbon atoms and a. Three vinylic hydrogens and two allylic hydrogens. b. Three vinylic hydrogens and one allylic hydrogen. c. Three vinylic hydrogens...
-
The two alkenes 2,3,3-trimethyl-1-butene and 1-octene were each subjected to allylic halogenation with N-bromosuccinimide. One of these alkenes yielded a single allylic bromide, whereas the other...
-
What are the premises for successful paleostress analysis?
-
Calculating components of cash outflow from operations Refer to Exhibit 5.22, which provides items from the financial statements of Information Technologies. a. How much cash did Information...
-
The following ledger accounts are for the Tuner Service Company: a. Cash b. Wages Expense c. Accounts Receivable d. R. Shuckman, Capital e. Service Revenue f. Prepaid Rent g. Accounts Payable h....
-
What was the approximate activity of the plutonium source at the start of the mission? A. \(2 \times 10^{21} \mathrm{~Bq}\) B. \(2 \times 10^{19} \mathrm{~Bq}\) C. \(2 \times 10^{17} \mathrm{~Bq}\)...
-
Roger Rosich saved $300,000 during the 30 years that he worked for a major corporation. Now he has retired at the age of 60 and has begun to draw a comfortable pension check every month. He wants to...
-
Pick a very specific item of clothing (not shoes or sneakers, not handbags, not cosmetics) that you regularly purchase. Then identify six factors that would cause industry supply for that item to...
-
What product(s) are expected when each of the following compounds reacts with one equivalent of NBS in CCl 4 in the presence of light and peroxides? Explain your answers. (a) cyclohexene (b)...
-
Predict the order of relative reactivities of the compounds within each series in S N 1 solvolysis reactions, and explain your answers carefully. (a) (b) Cl T -CH- CH3 (1) CH3 of CH3 (1) -C-Cl CHO...
-
Sketch the graph of y = sin in the interval 90 270.
-
How does an expected insurance reimbursement for a business casualty loss affect the amount of a casualty loss deduction? Does it make a difference whether an expected insurance reimbursement for...
-
Bevis Bag Co. purchased a tract of land on August 31 of the current year, paying $225,000. Prior to the sale, the seller paid property taxes of $18,000 on the property. The taxes covered the calendar...
-
Put yourself in the position of a management trainer. You have been asked to make a short presentation to the local Business Improvement Association at its biweekly luncheon. The topic you are to...
-
Don Smith's wife died in January while still employed and, as her beneficiary, he began receiving an annuity of $147 per month. There was no investment in the contract after June 30, 2013. The...
-
At the beginning of the year, NRD Company purchased the rights to a natural resource for $10,000,000. The estimated recoverable units from the natural resource amount to 3,500,000 units. During the...
-
A manufacturing company produces plastic pipes that are specified to be 10 inches long and 1/8 inch thick with an opening of 3/4 inch. These pipes are molded on two different machines. To maintain...
-
If the joint cost function for two products is C(x, y) = xy2 + 1 dollars (a) Find the marginal cost (function) with respect to x. (b) Find the marginal cost with respect to y.
-
Give the structure of each of the following compounds. (In some cases, more than one correct answer is possible.) (a) A six-carbon alkene whose proton NMR spectrum consists of one singlet (b) A...
-
Give the structure that corresponds to each of the following molecular formulas and NMR spectra: (a) C5H10; 1.5, s (b) C2H2F3I: 3.56 (q, J = 10 Hz) (c) C6H14O: 0.91 (6H, d, 7 = 7 Hz); 1.17(6H,...
-
A compound A reacts with H2 over Pd/C to give methylcyclohexane. A colleague, A1 Keen, has deduced that the compound must be either 1-methylcyclohexene or 3-methylcyelohexene. You have been called in...
-
M1 = (. ) , M2 (1 - ) 1. Compute the eigenvalues of the following matrices, and for each eigenvalue, find an associated eigenvector 3 -5 M3 = (*)) and M4 = (C) 2. Given the matrix equations dY;(t) A....
-
If a Department Manager is hired to strictly directly assist the clinic in its routine operations and making $6,000 per month, is that a direct expense?
-
4) Calculate the coordinates of the points labeled 1 and 2 on the line connecting points T and M, as well as the coordinates of point P, which is located perpendicular to point 1, the given...

Study smarter with the SolutionInn App


