Given that the stretching vibration of a typical CH bond has a frequency of about 9 *
Question:
Given that the stretching vibration of a typical C—H bond has a frequency of about 9 * 1013 s–1, which peak(s) in the IR spectrum of nonane (Fig. 12.4) would you assign to the C—H stretching vibrations?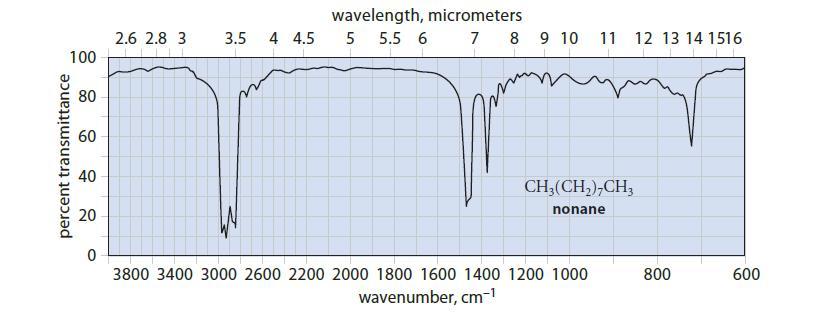
Transcribed Image Text:
percent transmittance 100 80 60 40 20 0 2.6 2.8 3 3.5 4 4.5 wavelength, micrometers 5 5.5 6 7 8 9 10 11 CH₂(CH₂)7CH3 nonane 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm-¹ 12 13 14 1516 800 600
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
From Eq 128a we kn...View the full answer

Answered By

Mahesh G
I have more than 7 years of experience in teaching physics, mathematics and python programming to more than 600 students including both online and offline tutoring.
I follow the following 7 step fundamental approach towards tutoring.
1. Curiosity, scope, enlightenment of the topic in hand.
2. Problem Definitions and elaboration.
3. Requisite mathematics, analytical abilities and quantitative
aptitude.
4. Preparing Algorithms for problem statement.
5. Concepts with analogies and building algorithm.
6. Introspection and improvising.
7. Daily class wise Cheat sheets(its not cheating) for consolidation.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which would be expected to be more intense, the stretching vibration of a C==O bond or the stretching vibration of a C==C bond?
-
Given that the force constants are similar for C--H and C--C bonds, explain why the stretching vibration of a bond occurs at a greater wavenumber.
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
You mentioned that Amazon has sustainability topics within us supply chain. How we deal with the criticism that Amazon itself tribute to overconsumption that's antithetical to sustainability
-
Mel Storrer believes there are no significant differences in the flow of costs between job order cost accounting and process cost accounting. Is Storrer correct? Explain.
-
Use Books-A-Millions financial statements, which can be found in Appendix A at the back of the book, to answer the following questions: 1. What types of debt does Books-A-Million have? Where did you...
-
On January 2, 2018, The Register, a science and technology news site based in Britain, published an article revealing the existence of two major bugs leaving virtually all computers and smartphones...
-
On January 1, 2014, Paloma Corporation exchanged $1,710,000 cash for 90 percent of the outstanding voting stock of San Marco Company. The consideration transferred by Paloma provided a reasonable...
-
A young child pushes her stuffed plush cow down a frictionless slide at the playground. The slide has a height h. The cow exits the slide horizontally at the bottom. The cow slides along the level...
-
One of the spectra in Fig. 12.11 is that of trans-2-heptene and the other is that of 2-methyl-1-hexene. Which is which? Explain. 100 percent transmittance (a) 80 60 40 0 2.6 2.8 3 3.5 4 4.5...
-
The base peak in the mass spectrum of 2,2,5,5-tetramethylhexane (molecular mass = 142) is at m/z = 57, which corresponds to a composition C 4 H 9 . (a) Suggest a structure for the fragment that...
-
Use the Binomial Theorem to expand and then simplify the result: (x 2 + x + 1) 3 .
-
A trader buys 200 shares of a stock on margin. The price of the stock is $20 per share. The initial margin is 60% and the maintenance margin is 30%. How much money does the trader have to provide...
-
Suppose that a bank has made a large number of loans of a certain type. The oneyear probability of default on each loan is 1.2%. The bank uses a Gaussian copula for time to default. It is interested...
-
An investor owns 10,000 shares of a particular stock. The current market price is $80. What is the worstcase value of the portfolio in six months? For the purposes of this question, define the...
-
Draw an E-R diagram for each of the following situations: a. A laboratory has several chemists who work on one or more projects. Chemists also may use certain kinds of equipment on each project....
-
A restaurant chain has several store locations in a city (with a name and zip code stored for each), and each is managed by one manager. Managers manage only one store. Each restaurant location has...
-
How much taxable income should each of the following taxpayers report? a. Kimo builds custom surfboards. During the current year, his total revenues are $90,000, and he incurs $30,000 in expenses....
-
When is the indirect pattern appropriate, and what are the benefits of using it?
-
Hemoglobin has pI = 6.8. Does hemoglobin have a net negative charge or net positive charge at pH = 5.3? At pH = 7.3?
-
Show how you could prepare the following -amino acids from the appropriate carboxylic acids: (a) Phenylalanine (b) Valine
-
What alkyl halides would you use to prepare the following -amine acids by the amidomalonate method? (a) Leucine (b) Histidine (c) Tryptophan (d) Methionine
-
A company pays $1200 plus 8% tax for a one-year insurance policy on June 1, 2021. How much is its insurance expense for the year ended December 31, 2021? Enter your answer without $ signs, decimal...
-
The Importance of Depreciation, Impairment, and Disposition from a Business Perspective All of the following statements about depreciation accounting are true EXCEPT one. Identify that one. Select...
-
Zain Hakim's client does not have in place appropriate controls for a risk he has identified. What should Zain do?

Study smarter with the SolutionInn App


