Given the boiling point of the first compound in each set, estimate the boiling point of the
Question:
Given the boiling point of the first compound in each set, estimate the boiling point of the second.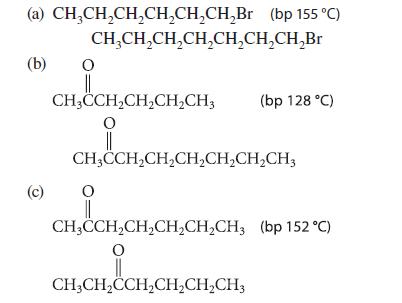
Transcribed Image Text:
(a) CH₂CH₂CH₂CH₂CH₂CH₂Br (bp 155 °C) (b) (c) CH₂CH₂CH₂CH₂CH₂CH₂CH₂Br O CH₂CCH₂CH₂CH₂CH3 2CH₂CH O || CH3CCH₂CH₂CH₂CH₂CH₂CH3 O (bp 128 °C) CH3CCH₂CH₂CH₂CH₂CH3 (bp 152 °C) O CH3CH₂CCH₂CH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Use the rule of thumb that an additional carbon ad...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
TIME 1 2 8 AM The following data were collected on the amount of deodorant in cans, in grams, filled in the filling line of a packaging company. Prepare X- and R-charts for the data, and comment on...
-
Estimate the boiling point of a 0.050 m aqueous MgCl2 solution. Assume a value of i based on the formula.
-
Glorious Electrical Appliances (GEP) Co. is a company that sells electrical tools. GEP uses perpetual inventory system in recording its inventory. The financial position of GEP as at 31 December 2016...
-
The Federal Reserve may raise its benchmark interest rate later this month. How is this achieved? Why would they do this? Explain the consequences fully. (Include graphs with your answer)
-
Multiple Choice 1. Colorado passes a hotel tax of 8 percent for Colorado residents and 15 percent for out-of-state visitors. The new law A. Is valid, based on the Supremacy Clause B. Is void, based...
-
Zaro Companys balance sheets for December 31, 2011 and 2010, income state-ment for the year ended December 31, 2011, and the statement of cash flows for the year ended December 31, 2011, follow: The...
-
The following selected accounts and balances appear on the adjusted trial balance for Ray Service, Inc., on December 31, 2010: 1. What is the net income or net loss? 2. What is the change in Retained...
-
A small vessel was purchased by a chemical company for $55,000 and is to be depreciated by MACRS depreciation . When its requirements changed suddenly, the chemical company leased the vessel to an...
-
Rob is saving for retirement immediately and wants to retire in 43 years. He will make an annual contribution into his retirement account. The contribution will be made at the end of each year. He...
-
Draw the structures and give the names of all isomers of octane with (a) Five carbons (b) Six carbons in their principal chains.
-
Give a general balanced reaction for (a) The complete combustion of an alkane (formula C n H 2n+2 ). (b) The complete combustion of a cycloalkane containing one ring (formula C n H 2n ).
-
Choose two stocks from different industries that you think would have a low correlation. Get the closing prices for each month over the past five years for both stocks. To get the prices from Yahoo!...
-
Continental Breakfast Company was formed in 2023. During the year the company had the following events occur: 1. The company issued $100,000 in common shares for cash. 2. The company purchased...
-
Case Scenario Hazel Blue Pty Ltd (HBPL) is an innovative research institute based in New South Wales, holding ownership of 30 patents as part of its intellectual property portfolio. The company's...
-
2. Explain in your own words how double entry accounting relates to the accounting equation. 3. Why is it important for businesses to prepare accounts reports periodically? Explain in your own words....
-
How do hormones modulate immune function and inflammatory responses, and what are the implications of hormone-immune interactions for autoimmune diseases, allergic reactions, and host-pathogen...
-
Cash sales are 4 0 percent in a given monin, with the remainder going into accounts receivable. All receivables are cotected in the monin tollowing tne sale, Esquire sells all or its goods for $ 2...
-
Dividend yield is the annual dividend paid by a company expressed as a percentage of the price of the stock (Dividend/Stock Price 3 100). The dividend yield for the Dow Jones Industrial Average...
-
Air pollution generated by a steel mill is an example of a) a positive production externality. b) a negative production externality. c) a public good. d) the free-rider problem. State and local taxes...
-
Predict the products of the reaction of (i) phenyl acetaldehyde and (ii) acetophenone with the following reagents: (a) NaBH4 then H3O+ (b) Tollens reagent (c) NH2OH, HC1 catalyst (d) CH3MgBr, then...
-
How would you prepare the following substances from 2-cyclohexcnone? More than one step may be required. .CH (a) (b) (d) (c) CSH5 (Two ways)
-
Show how the Wittig reaction might be used to prepare the following alkenes. Identify the alkyl halide and the carbonyl components that would he used. (b) (a)
-
Suppose a sample of a certain substance decayed to 73.2% of its original amount after 300 days. (Round your answers to two decimal places.) (a) What is the half-life (in days) of this substance? days...
-
Estimate the distance you can travel in 7 hours 55 mins if u drive an average of 41 miles per hour. round to the mile
-
For the function f(x) = 1-x and the partition of the interval [-1,2] given by P = {-1,-1,1,2}: a. Make a sketch of this function on the indicated interval. (3 pts) b. What is n, the number of...

Study smarter with the SolutionInn App


