Identify the following compounds. (a) C0H0O NMR: 8 2.82 (6H, s), 8 8.13 (4H, s) IR: 1681
Question:
Identify the following compounds. 
Transcribed Image Text:
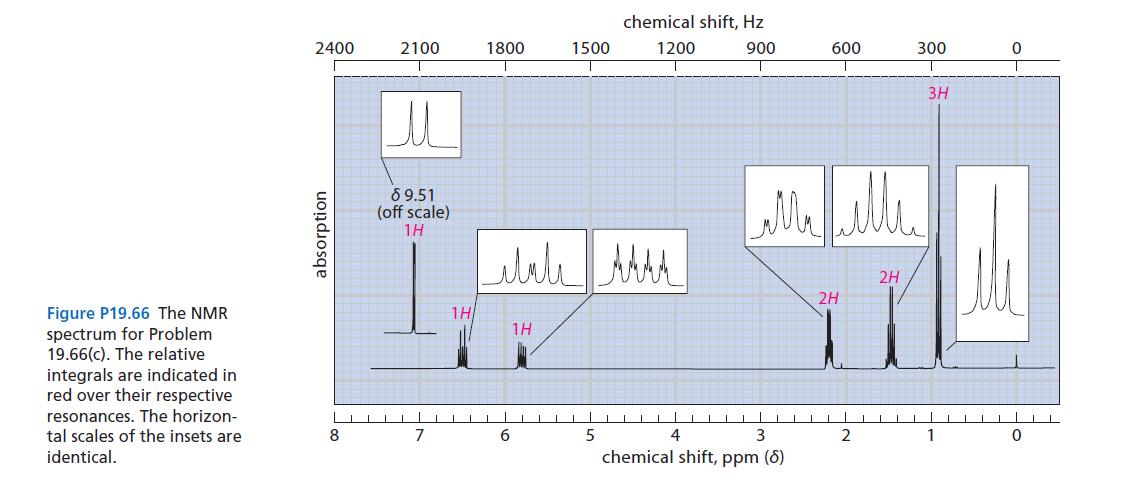
(a) C₁0H₁0O₂ NMR: 8 2.82 (6H, s), 8 8.13 (4H, s) IR: 1681 cm-¹, no 0-H stretch (b) C-H₁0ONMR: 8 9.8 (1H, s), 8 1.1 (9H, s) NMR in Fig. P19.66 (p. 1002) IR: 1701 cm-¹, 970 cm-¹ (c) CH₁0O 10 UV: Amax = 215 (€ = 17,400), 329 (€ = 26)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The NMR spectrum suggests a great degree of symmetry four aromatic protons all equivalent and two ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the following compounds on the basis of the information provided: (a) C9H12O: Its infrared and 1H NMR spectra are shown in Figure 24.5. (b) C9H11BrO: Its infrared and 1H NMR spectra are...
-
Identify the following compounds as being a hydrocarbon, an alcohol, an ether, or a carboxylic acid. (a) (b) (c) (d)
-
Identify the following compounds from their IR and proton NMR spectra. (a) C4H6O: liberates a gas when treated with C2H5MgBr (b) C5H6O IR: 3300, 2102, 1634 cm-1 NMR: 3.10 (1H, d, J = 2Hz); 3.79 (3H,...
-
The preferred stock of Walter Industries Inc. currently sells for $36 a share and pays $2.50 in dividends annually. What is the firms cost of capital for the preferred stock?
-
The adjusted trial balance from the January worksheet of Silver Sign Company follows: Requirements 1. Journalize Silvers closing entries at January 31. 2. How much net income or net loss did Silver...
-
An article in The Wall Street Journal (July 11, 2008) outlined a number of reasons as to why the 16 teams in Major League Baseballs National League (NL) are inferior to the 14 teams in the American...
-
Show that after nearly all of the positrons were annihilated and the electron number density had nearly leveled off at the proton density, the ratio of the positron number density to the photon...
-
Hallas Company manufactures a fast-bonding glue in its Northwest plant. The company normally produces and sells 40,000 gallons of the glue each month. This glue, which is known as MJ-7, is used in...
-
Define the relational model? What does the relational model consist of? What are the 4 relational models?Discuss
-
Identify the compound with the mass spectrum and proton NMR spectrum shown in Fig. P19.67. This compound has IR absorptions at 1678 cm 1 and 1600 cm 1 . 100 183 185 155 60 40 1157 43 198 200 La fl 0...
-
Give curved-arrow mechanisms for the reactions given in Fig. P19.64. (b) (c) S PhCPh (CH3O) 3P: + HC-CH-CH3 CH,CH,NH, + (d) H CH3 HO CH O Figure P19.64 Ph-C-Ph + HS HCI O=CH CH O acid H3C CH3 H CH3...
-
A small computer manufacturing company produces both laptop and desktop computers. The production of a laptop computer requires 4 hours of assembly and 1 hour of testing time. The production of a...
-
What is the output of the following code: #include int main() { } int x = 12; do { printf("Hello World "); --X; } while ( x 5 == 0); return 0; How many times " Hello World " will be printed?
-
In the stone age when I was in grad school 2000s, Kia was trying to get into the US market or at least expand to the southeast. They didn't have the best reputation as quality goes. Some of you may...
-
The following information relates to the Kevin Hall Company. Date Ending Inventory (End-of-Year Prices) Price Index December 31, 2016 $ 63,200 100 December 31, 2017 120,384 132 December 31, 2018...
-
A manufacturing company applies factory overhead based on direct labor hours. At the beginning of the year, it estimated that factory overhead costs would be $482,910 and direct labor hours would be...
-
The following transactions occurred in the order shown in 2019 for a new firm called ACCO Ltd. (The fiscal year is the same as the calendar year.) I. II. III. IV. V. VI. Received $8,400,000 cash from...
-
What are the purposes of a product-costing system?
-
The Heese Restaurant Group manufactures the bags of frozen French fries used at its franchised restaurants. Last week, Heeses purchased and used 101,000 pounds of potatoes at a price of $ 0.70 per...
-
Show how you might use thioacetal formation and Raney nickel desulfurization to convert: (a) cyclohexanone to cyclohexane and (b) benzaldehyde to toluene.
-
(a) Show how you might prepare lactic acid from acetaldehyde through a cyanohydrin intermediate. (b) What stereoisomeric form of lactic acid would you expect to obtain? OH CO2H
-
In addition to triphenylphosphine, assume that you have available as starting materials any necessary aldehydes, ketones, and organic halides. Show how you might synthesize each of the following...
-
2. Let R be the relation {(1,2), (1,3), (2,3), (2,4), (3,1)), and let S be the relation ((2,1),(3,1), (3,2), (4,2)) Find SoR.
-
Consider a Playfair cipher that uses the password "WORKHARD". 1. Construct the matrix associated with this Playfair cipher. 2. Encrypt the plaintext message: "EVENINGS". Show your work step by step....
-
III. a) Apply the subnet mask 255.255.252.0 to the IP address 192.105.103.211 b) Apply the subnet mask 255.255.255.192 to the IP address 131.200.105.145

Study smarter with the SolutionInn App


