Isoniazid is an antituberculosis drug that operates by reacting with pyridoxal phosphate in the causative Mycobacterium. Show
Question:
Isoniazid is an antituberculosis drug that operates by reacting with pyridoxal phosphate in the causative Mycobacterium. Show how isoniazid reacts with pyridoxal phosphate.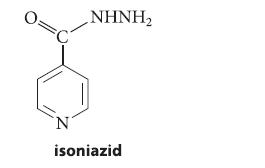
Transcribed Image Text:
.NHNH, N isoniazid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Isoniazid is a type of amide called a hydrazide Semicarbazide Table 193 text p 985 is the same type ...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Behind terrorism is a complex endeavor, shaped by various factors and objectives unique to each terrorist group. In Max Abrahms article, "What Terrorists Really Want: Terrorist Motives and...
-
Pyridoxal phosphate (PLP) is a form of vitamin B 6 . PLP serves as a coenzyme in the enzyme-catalyzed decarboxylation of meso-2,6-diaminopimelic acid, which is thelast step in the biosynthesis of the...
-
The racemization of amino acids is an important reaction in a number of bacteria. This is a pyridoxal-phosphate catalyzed reaction. Outline a curved-arrow mechanism for this reaction showing clearly...
-
Use Eq. (6) to estimate Af = f(3.53, 8.98) - f(3.5,9)
-
What criteria must be met before donated services can be recorded as contribution revenue and an expense? Give an example of a service that might qualify as a donated service for accounting purposes.
-
Fred Jackson, president of Bailey Company, is concerned about the companys ability to obtain a loan from a major bank. The loan is a key factor in the firms plan to expand its operations. Demand for...
-
What is the speed of an electron after being accelerated from INT rest through a \(20 \times 10^{6} \mathrm{~V}\) potential difference?
-
The Home Shopping Club ordered 12,000 Care Bear lamps from Ohio International, Ltd. When the lamps arrived, they had poor painting and staining, elements were improperly glued and could come loose (a...
-
In ACFR, please explain Concentration of credit risk represents the risk of investments in any one issue that represents five percent or more of investments. The City's investment policy limits the...
-
Which compound should readily undergo substitution of the bromine by phenolate anion: 4- bromopyridine or 3-bromopyridine? Explain, and give the structure of the product.
-
Give the structure of the product and a curved-arrow mechanism for its formation in the reaction of 4-chloropyridine with sodium methoxide. Draw all important resonance structures for the addition...
-
This histogram shows the average annual premium (in dollars) for homeowners insurance paid in the 50 states and District of Columbia.19 (a) Which holds more states, the interval from $600 to $700 or...
-
The following case highlights the right of the taxpayer to select among legitimate business alternatives in order to avoid taxes. Read the case and prepare a written brief.
-
What is the distinction between deductions for adjusted gross income and deductions from adjusted gross income?
-
When are capital expenditures incurred for medical reasons deductible?
-
Can a taxpayer qualify for the household and dependent care credit if he or she is not employed?
-
Why can't a partner recognize loss upon receipt of a current distribution?
-
Wisconsin is an important milk-producing state. Some people might argue that because of transportation costs, the cost of milk increases with the distance of markets from Wisconsin. Suppose the milk...
-
On average there are four traffic accidents in a city during one hour of rush-hour traffic. Use the Poisson distribution to calculate the probability that in one such hour there arc (a) No accidents...
-
Which one of the following stereoisomers should undergo β-elimination most rapidly with sodium ethoxide in ethanol? Explain your reasoning. CH1 CH3 Br CH3 CH
-
The reaction of butylamine, CH3(CH2)3NH2, with 1- bromobutane rn 60Vo aqueous ethanol follows the rate law rate = k[butylamine] [1 -bromobutane] The product of the reaction is (CH3CH2CH2CH2)2NH2+,...
-
Consider the reaction sequence given in Fig. p9.70. (Bu- - butyl group : CH3CH2CH2CH2 .) (a) Use what you know about the stereochemistry of bromine addition to propose the stereochemistry of compound...
-
Based on the materials provided, how (if at all) does the GIM investment methodology differ from the approach used by traditional value investors (i.e., a strategy of buying when the intrinsic value...
-
1 2 3 Revenue 5 6 Total Revenue (Credit) 7 Cost of Revenue 8 Gross Profit 10 Operating Expenses B D AMAZON.COM (AMZN) INCOME STATEMENT (all numbers are in thousands) 12/31/18 12/31/17 G H J Required:...
-
What is the income tax formula and what each item of the formula means.? ) What is gross income?

Study smarter with the SolutionInn App


