Noting the LX character of the allyl ligand in Table 18.1, sketch the allylmetal interaction, showing both
Question:
Noting the LX character of the allyl ligand in Table 18.1, sketch the allyl–metal interaction, showing both L-type and X-type bonds. Use M as a general metal.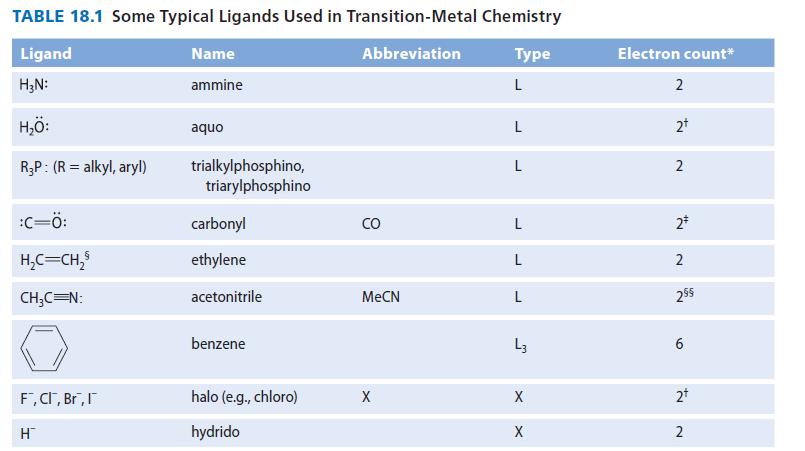
Transcribed Image Text:
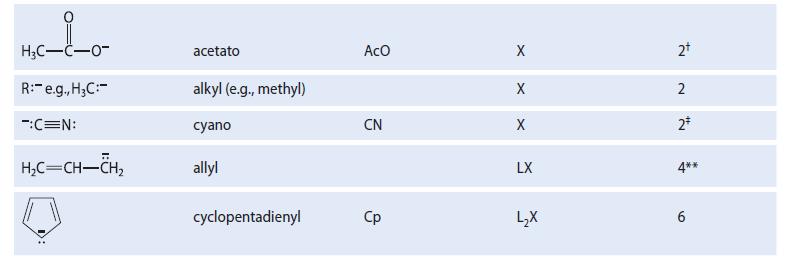
TABLE 18.1 Some Typical Ligands Used in Transition-Metal Chemistry Abbreviation Ligand H₂N: H₂O: R3P: (R = alkyl, aryl) :c=0: H₁₂C=CH₂5 CH₂C=N: F, Cl, Br, I H™ Name ammine aquo trialkylphosphino, triarylphosphino carbonyl ethylene acetonitrile benzene halo (e.g., chloro) hydrido со MeCN X Type L L L L L L L3 X X Electron count* 2 2+ 2 2* 2 295 6 2+ 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The bond to one allylic carbon is an Xtype bond That is if we break the me...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Emily Jackson (Social Security number 765-12-4326) and James Stewart (Social Security number 466-74-9932) are partners in a partnership that owns and operates a barber shop. The partnership's first...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Modify Lookup to make a program LookupAndPut that allows put operations to be specified on standard input. Use the convention that a plus sign indicates that the next two strings typed are the...
-
(a) In the reference frame of the muon in Problem 4, how far does the laboratory travel in a typical lifetime of 2 s? (b) What is this distance in the laboratorys frame?
-
At the end of 2007 the Ritter Company prepared a trial balance, recorded and posted its adjusting entries, and then prepared an adjusted trial balance. Selected accounts and account balances from the...
-
What factors provide an upper bound on the period at which the system timer interrupts for preemptive context switching?
-
Diagram the sequence in which the following source document is prepared. a. Bill of Materials b. Work order c. Sales forecast d. Materials requisition e. Move ticket f. Production schedule g. Route...
-
How well does your organization accommodate the three phases of transition? Which phase is the most challenging for you during a period of transition? 2. What are the key strategies that help people...
-
Which of the two compounds in each of the following sets should react more rapidly in a nucleophilic aromatic substitution reaction with CH 3 O in CH 3 OH? Explain your answers. (a) NO or F NO (b) NO...
-
Within each series, arrange the compounds according to increasing rates of their reactions by the S N 1E1 mechanism. Explain your reasoning. (b) Br -C=CH, A Cl -CH-CH3 A Br | -CH-CH3 B B Br C Cl...
-
Toro Co. has equipment with a carrying amount of $700,000. The value-in-use of the equipment is $705,000, and its fair value less costs of disposal is $590,000. The equipment is expected to be used...
-
One of the issues that is debated in virtually every election is whether to raise the federal minimum wage, which stood at $7.25 per hour in 2018. Suppose that you are married with a child, working a...
-
How would you expect the possibility of differentiating your product to change competition between oligopolists?
-
The 2018 federal poverty guidelines used to determine eligibility for Medicaid and the Childrens Health Insurance Program (CHIP) set a poverty line of $25,100 for a family of four in the 48...
-
As corruption falls in a country, cost of production often falls. Why?
-
Conduct an online search for iPhone repair San Diego You will find about 10 million entries. Now try iPhone screen repair San Diego. You will find more than 6 million entries. Try to estimate the...
-
A company produces and ships 16 personal computers knowing that 4 of them have defective wiring. The company that purchased the computers is going to thoroughly test three of the computers. The...
-
The value of a share of common stock depends on the cash flows it is expected to provide, and those flows consist of the dividends the investor receives each year while holding the stock and the...
-
(a) Using bond-dissociation enthalpies from Table 4-2 (page 143), calculate the heat of reaction for each step in the free-radical bromination of methane. (b) Calculate the overall heat of reaction....
-
The reaction of tert-butyl chloride with methanol Is found to follow the rate equation Rate = kf [(CH3)3C-Cl] (a) What is the kinetic order with respect to tert-butyl chloride? (b) What is the...
-
Under certain conditions, the bromination of cyclohexene follows an unusual rate law: Rate = kr [cyclo-hexene][Br2]2 (a) What is the kinetic order with respect to cyclo-hexene? (b) What is the...
-
discuss the role of viruses in driving evolutionary processes, including their impact on host genome evolution, genetic diversity of host populations, and co-evolutionary dynamics between viruses and...
-
A customer walks into your jewelry store with printouts of diamond selections from Blue Nile, a company that is the largest online retailer of diamonds. The list price for the customer's desired...
-
Can you provide a significant historical event in the evolution of Supply Chain Management, and identify a key driver in the evolution of Supply Chain Management form (1900 - 1910 ) or (1935 to 1945)...

Study smarter with the SolutionInn App


