Outline a sequence of reactions by which d-glucose can be converted into methyl 2,3,4,6-tetra-O-acetyld-glucopyranoside. ACO ACOCH ACO-
Question:
Outline a sequence of reactions by which d-glucose can be converted into methyl 2,3,4,6-tetra-O-acetyld-glucopyranoside.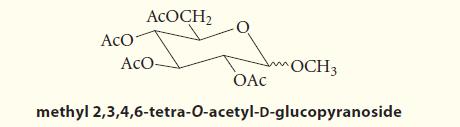
Transcribed Image Text:
ACO ACOCH₂ ACO- OCH 3 OAC methyl 2,3,4,6-tetra-O-acetyl-D-glucopyranoside
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
In solving problems of this sort in which apparently similar hydroxy groups are converte...View the full answer

Answered By

Moses mwangi
With prior writing experience, be sure that I will give a great grade, If not an A+, it will be something close to this. My reviews speaks it all, Try me!!
4.80+
78+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a sequence of reactions that would bring about the conversion of aniline into each of the following compounds. (a) Benzyl alcohol (b) N-phenyl-2-butanamine
-
Outline a sequence of reactions that will bring about each of the following conversions. o-glucopyranose to 2,3,4,6-tetra-O-benzyl-o-glucopyranose
-
(A) Using Figure 21-19, write chemical equations for the sequence of reactions by which borax is converted to diborane. (B) Using Figure 21-19, write chemical equations for the sequence of reactions...
-
Given a sorted array of Comparable items, write functions floor () and ceiling () that return the index of the largest (or smallest) item not larger (or smaller) than an argument item in logarithmic...
-
The rest energy of a proton is about 938 MeV. If its kinetic energy is also 938 MeV, find (a) Its momentum (b) Its speed.
-
Peabody & Peabody has 2012 sales of $10 million. It wishes to analyze expected performance and financing needs for 20142 years ahead. Given the following information, respond to parts a and b. (1)...
-
A higher level of hemoglobin in the blood increases the bloods density. This is the basis for a simple test that can be used to see if a prospective blood donor has a high enough hemoglobin level to...
-
Meyer reported the following pretax financial income (loss) for the years 20062010. 2006 .....$240,000 2007 .......350,000 2008 .......120,000 2009 ......(570,000) 2010 .......180,000 Pretax...
-
Barker Company had reported the following details for the year ended December 31, 2025: Net sales Total assets Total liabilities $25,650,000 17,300,000 10,600,000 The company's beginning total assets...
-
Name each of the following aldoses. In part (a), work back to the Fischer projection and consult Fig. 24.3. In part (b), decide which carbons have configurations different from those of glucose, and...
-
Convert the Fischer projection of -d-glucopyranose into a Haworth projection, a line-and-wedge structure, and a chair conformation. (A Haworth projection is defined below.)
-
A procedure often used to control the quality of name-brand food products utilizes a panel of five "tasters." Each member of the panel tastes three samples, two of which are from batches of the...
-
How do the traditional, new Keynesian, and real business cycle models differ in their views about the efficacy of discretionary policy?
-
How does the theory of rational expectations differ from that of adaptive expectations?
-
In the new Keynesian model, what shocks cause business cycle fluctuations? Does it matter whether these shocks are anticipated or unanticipated? Explain.
-
Assume that the expenditure and tax multipliers can be estimated at 0.75 and 0.5, respectively. a) Would you recommend expansionary fiscal policy based on tax cuts or increased government...
-
How and why do changes in the real interest rate affect net exports?
-
Suppose the annual employer 401(k) cost per participant is normally distributed with a standard deviation of $625, but the mean is unknown. a. If 73.89% of such costs are greater than $1,700, what is...
-
Bonus shares can be issued out of revenue reserves. True/False?
-
Label the reactants in these acid-base reactions as Lewis acids (electrophiles) or Lewis bases (nucleophiles). Use curved arrows to show the movement of electron pairs in the reactions. (a) (b) (c)...
-
Predict the products of the following acid-base reactions. (a) (b) (c) (d) (e) (f) (g) (h) CH,COOH + (CH, ),N : H2O + NH3 HCOOH + CH,O- NH,CH,COOH 2 OH
-
The following compounds are listed in increasing order of acidity. In each case, the most acidic proton is shown in red. (a) Show the structure of the conjugate base of each acid, including any...
-
The Self-Reflection Essay will reflect upon your experiences at your employment site during this internship course and what you have learned. Include how these experiences may have furthered your...
-
A law firm with three partners is considering going completely paperless and to a cloud solution. This is being pushed primarily by one partner, and his challenge, now that he has been given the...
-
An investment project offers $4,350 per year forever, with the first payment occurring one year from now. If the interest rate is 6%, what would you pay to participate in this project? Hint: this is...

Study smarter with the SolutionInn App


