Outline a synthesis of each of the following compounds from mesityl oxide (4-methyl-3-penten-2-one). Use an organometallic reagent
Question:
Outline a synthesis of each of the following compounds from mesityl oxide (4-methyl-3-penten-2-one). Use an organometallic reagent in at least one step of each synthesis.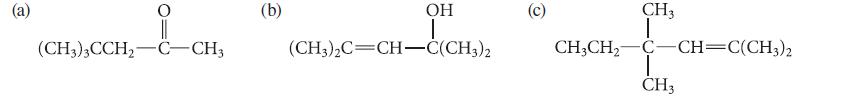
Transcribed Image Text:
i (CH3)3CCH₂-C-CH3 (b) OH I (CH3)2C=CH-C(CH3)2 (c) CH3CH₂ CH 3 I C C—CH=C(CH3)2 T CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a b c CH3CCHCCH 3 0 mesityl oxide 4methyl3penten2one ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) Benzyl methyl ether from toluene (b) (c) (d) CH2CH O from cyclopentene 7-CO.H...
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) 1-cyclohexyl-2-methyl-2-prupanol from bromocyclohexane (b) PhNHCH2CH2CH(CH3)2...
-
Outline a synthesis of each of the following compounds from a -keto ester; then show how the -keto ester itself can be prepared. CHj
-
Broadmore Corporation acquired 75 percent of Stem Corporations common stock on January 1, 20X8, for $435,000. At that date, Stem reported common stock outstanding of $300,000 and retained earnings of...
-
Review the data in Exercise 6-21. Requirements 1. Compute the cost of goods sold under FIFO. 2. Compute the cost of goods sold under LIFO. 3. Which method results in the higher cost of goods sold?
-
You are considering an investment in 30-year bonds issued by Moore Corporation. The bonds have no special covenants. The Wall Street Journal reports that one-year T-bills are currently earning 3.25...
-
A piece of cloth is discovered in a burial pit in the southwestern United States. A tiny sample of the cloth is burned to CO 2 , and the 14 C/ 12 C ratio is 0.250 times the ratio in todays...
-
The scal year of Duchess County ends on December 31. Property taxes are due March 31 of the year in which they are levied. 1. Prepare journal entries (excluding budgetary and closing entries) to...
-
x-4 + 3x+21 Fully simplify the NPV's. x-8x+16 x2 +12x+35 expression below. Don't forget to list your
-
Which of the following is something that an interviewee might do or feel while in the denial phase? a. Insult, harm, or slander. b. Attempt to justify the act. c. Become sad, withdraw, or lose...
-
Show how ethyl 2-butenoate can be used as a starting material to prepare (a) Ethyl butanoate and (b) 2-buten-1-ol.
-
Give some examples of systems analysis decisions that involve a trade-off between each of the following pairs of objectives: a. Economy and usefulness b. Economy and reliability c. Economy and...
-
You work for a company, which expects to earn at least 7 percent on its projects. The cash flow information for 4 potential projects is described below. Which of the projects would you fund if the...
-
What 3 options do you have when allocating employee wages to expense accounts?
-
Sony and Loews In April 1998, officials at Sony were ecstatic. The Department of Justice (DOJ) had agreed to allow their theater division to acquire a substantial interest in Cineplex Odeon. Under...
-
Read the extract, which is about a universal basic income. Then, drawing upon module material as well, answer the following three questions. 3.1 Outline what is meant by a universal basic income and...
-
1. In "The Blessing of SoccerLinks to an external site.," what is the specific historical reason Enrique Krauz hypothesizes as to why soccer/ futbol is so popular in Mxico? 2. According to Joshua H....
-
During June, Danby Companys material purchases amounted to 6,000 pounds at a price of $7.30 per pound. Actual costs incurred in the production of 2,000 units were as follows: Direct labor:...
-
The words without recourse on an indorsement means the indorser is: a. not liable for any problems associated with the instrument. b. not liable if the instrument is dishonored. c. liable personally...
-
Compound S (C8H16) reacts with one mole of bromine to form a compound with molecular formula C8H16Br2. The broadband proton-decoupled 13C spectrum of S is given in Fig. 9.49. Propose a structure for...
-
A compound with molecular formula C4H8O has a strong IR absorption at 1730 cm-1. Its mass spectrum includes key peaks at m/z 44 (the base peak) and m/z 29. Propose a structure for the compound and...
-
In the mass spectrum of 2, 6-dimethyl-4-heptanol there are prominent peaks at m/z 87, 111, and 126. Propose reasonable structures for these fragment ions.
-
BuyCo, Inc., holds 29 percent of the outstanding shares of Marqueen Company and appropriately applies the equity method of accounting. Excess cost amortization (related to a patent) associated with...
-
Change the total fixed manufacturing overhead cost for the Milling Department in Data area back to $390,000, keeping all of the other data the same as in the original example. Consider a new job, Job...
-
Fickel Company has two manufacturing departments-Assembly and Testing & Packaging. The predetermined overhead rates in Assembly and Testing & Packaging are $12.00 per direct labor-hour and $8.00 per...

Study smarter with the SolutionInn App


