Rank the following compounds in order of increasing S N 2 reaction rate with KI in acetone.
Question:
Rank the following compounds in order of increasing SN2 reaction rate with KI in acetone.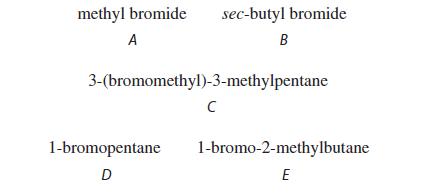
Transcribed Image Text:
methyl bromide sec-butyl bromide A B 3-(bromomethyl)-3-methylpentane с 1-bromopentane 1-bromo-2-methylbutane E D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The first thing to do is to draw out the structures The order of increasing S N 2 rea...View the full answer

Answered By

Muhammad Khurram
I have strong General Management skills to apply in your projects. Over last 3 years, I have acquired great knowledge of Accounting, Auditing, Microsoft Excel, Microsoft PowerPoint, Finance, Microsoft Project, Taxation, Strategic Management, Human Resource, Financial Planning, Business Planning, Microsoft Word, International Business, Entrepreneurship, General Management, Business Mathematics, Advertising, Marketing, Supply Chain, and E-commerce. I can guarantee professional services with accuracy.
4.80+
249+ Reviews
407+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank the following compounds in orcler of increasing SN2 reaction rate with KI in acetone. methyl bromide sec-butyl bromide 3-(bromomethyl)-3-methylpentane I-bromopentane -bromo-2-methylbutane
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Rank the following compounds in order of increasing λ max :
-
Prove that opposite sides of a quadrilateral circumscribing a circle subtend supplementary angles at the centre of the circle.
-
The Village of Budekville, which has a fiscal year July 1 to June 30, sold $3,000,000 in 6 percent tax-supported bonds at par to construct an addition to its police station. The bonds were dated and...
-
The total factory overhead for Bardot Marine Company is budgeted for the year at $ 600,000 divided into two departments: Fabrication, $ 420,000, and Assembly, $ 180,000. Bardot Marine manufactures...
-
If a well-behaved investment alternative's internal rate of return (IRR) is equal to MARR, which of the following statements about the other measures of worth for this alternative must be true? 1....
-
The cash account for Online Medical Co, at June 30, 2012, indicated a balance of $9,375. The bank statement indicated a balance of $10,760 on June 30, 2012. Comparing the bank statement and the...
-
2024 2023 2022 Sales $ 78,000 Cost of goods sold 62,400 $ 70,000 60,900 $ 59,000 44,100 2021 $ 58,000 35,100 2020 $ 50,000 30,000 Required: Dollar amounts stated are in thousands. a. Compute trend...
-
In each of the following series, order the atoms, compounds, or ions in order of increasing polarizability, and explain your choices. (a) Se, O, S (b) Chloroform, fluoroform, iodoform (c) I , Br ,...
-
Give the structure that meets the criteria given in each of the following cases: (a) A compound C 6 H 14 that gives only two products of monochlorination, one of which is chiral. (b) Four...
-
Find A B C if A = {x: 0 x 4}, B = {x: 2 x 6}, and C = {x: x = 0, 1, 2,...}.
-
Plato PLC is a supplier to large construction companies. During 2017, Plato experienced an increase in net fixed assets of 1,500,000 and had depreciation of 1,000,000. It also experienced an increase...
-
In what circumstances is frustration likely to generate destructive behaviour?
-
What is understood by the applicantorganization ft approach to the selection interview?
-
How is the price of petroleum determined and why is it so volatile?
-
York Engineering Ltd. forecasts sales of 1,250,000 for 2022. Assume that the firm has fixed costs of 320,000 and variable costs amount to 30% of sales. Operating expenses are estimated to include...
-
Dorothy & George Company is planning to acquire a new machine at a total cost of $30,600. The machines estimated life is six years and its estimated salvage value is $600. The company estimates that...
-
For the next several days, take notes on your listening performance during at least a half-dozen situations in class, during social activities, and at work, if applicable. Referring to the traits of...
-
Long before chiral allenes were known (Problem 9.72), the resolution of 4-methylcyclohexylideneacetic acid into two enantiomers had been carried out. Why is it chiral? What geometric similarity does...
-
(S)-i-Chloro-2-mcthylbutane undergoes light-induced reaction with Cl2 by a radical mechanism to yield a mixture of products, among which are 1, 4-dichloro-2-methylbutanc and 1,...
-
Draw the structure of a meso compound that has five carbons and three chirality centers.
-
what is the Lululemon Inventor valuation method?
-
What are factors to effect ( increase or decrease ) will the customers are given a large discount for cash transactions? the company discontinues the production of a product which has not sold for a...
-
Bridgeport Inc. incurred a net operating loss of $578,700 in 2023. Combined income for 2020, 2021, and 2022 was $457,000. The tax rate for all years is 30%. Assume that it is more likely than not...

Study smarter with the SolutionInn App


