Supply the curved-arrow notation for the acid- catalyzed isomerization shown in Fig. P4.64. H-SO,H HC CH3 HC
Question:
Supply the curved-arrow notation for the acid- catalyzed isomerization shown in Fig. P4.64.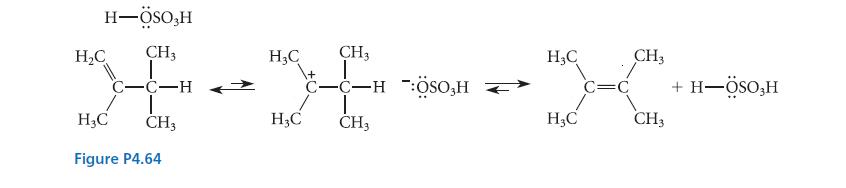
Transcribed Image Text:
H-ÖSO,H H₂C CH3 H₂C 24-34 C-C-H H3C CH3 H₂C CH3 Figure P4.64 CH3 C-C-H ÖSO,H H₂C H₂C C=C CH3 CH3 + H-ÖSO3H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The mechanism is a sequence of two Brnsted acidbase reactions In the first step ...View the full answer

Answered By

Emily Grace
With over a decade of experience providing top-notch study assistance to students globally, I am dedicated to ensuring their academic success. My passion is to deliver original, high-quality assignments with fast turnaround times, always striving to exceed their expectations.
4.90+
3+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the curved arrow notation and the product for the nucleophilic addition of a propyl anion to cyclohexanone. Draw the curved arrow notation Draw the product with lone pairs and nonzero formal...
-
It is known that the pressure and temperature of a storage tank that stores the cyclohexane vapor are 16.292 bar and 719.68 K. The universal gas constant, R, is given as 83.14 cm bar/(mol- K). Other...
-
Analyze the case study, "Frank Smith Plumbing." Analyze the "Frank Smith Plumbing's Financial Statement" spreadsheet. Compare the cost of the truck to the cash flow records Compile your calculations...
-
What are some ways that a manager can identify his or her knowledge of the feasible set of alternatives?
-
Prahm & Associates had EBIT of $5M last year. The firm carried an average debt of $15M during the year on which it paid 8% interest. The company paid no dividends and sold no new stock. At the...
-
Refer to the statements for Google in Appendix A. For the year ended December 31, 2015, what was its debt-to-equity ratio? What does this ratio tell us? Data From Statement Google In Appendix A...
-
Superior Company provided the following account balances for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses . . . . . . . . . . . . . . ....
-
What is the interest earned on $350 invested 4 years at a 5% simple interest?If I put $1500 into my savings account and earned $180 of interest at 4% simple interest, how long was my money in the...
-
The standard free energy of formation, G f , is the freeenergy change for the formation of a substance at 25 C and 1 atm pressure from its elements in their natural states under the same conditions....
-
Consider the following compounds and their dipole moments: Assume that the CCl bond dipole is oriented as follows in each of these compounds. (a) According to the preceding dipole moments, which is...
-
Problems 103110 are based on material learned earlier in the course. The purpose of these problems is to keep the material fresh in your mind so that you are better prepared for the final exam. Find...
-
2) Family Fun reported credit sales of $800,000, cash collections of $550,000 and bad debt expense of $15,000 for last year. Accounts receivable had a balance of $1,000,000 at the beginning of the...
-
If the future value of an ordinary, 7-year annuity is $9,000 and interest rates are 9.0 percent, what's the future value of the same annuity due? (Round your answer to 2 decimal places.)
-
Burke Tires just paid a dividend of Do=$1.3. Analysts expect the company's dividend to grow by 30% this year, by 10% in Year 2, and at a constant rate of 5% in Year 3 and thereafter. The required...
-
KL and LP are both engaged in retKL limited and LP limited are both engaged in retailing but they seem to take a different approach. one of them prides itself on personal service and the other on...
-
What are the differences between cash and accrual basis accounting? Provide an example of each. Accrual accounting requires adjusting entries. Provide an example of an adjusting entry.
-
The comparative balance sheets for 2011 and 2010 and the statement of income for 2011 are given below for Wright Company. Additional information from Wright's accounting records is provided also....
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
Draw structures for these compounds: (a) Diethyl ammonium bromide (b) N-Methyl-3-(1-methylpropyl)-2-octanamine
-
Name thesecompounds: CH3 CH3 T a) CHCH,CH_CHCHCH,CH, CHCH3 b) CHCH=CHCHCHCH3 CI c) CH,CH,CHC=CH d) e) f) g) OH CH3 H
-
Draw structures for these compounds: (a) 5-Ethyl-4-methylnonane (b) 2-Methyl-1, 3-hexadiene (c) 3-Methylcyclopentanol (d) 3-Octyne (e) sec-Butylcyclohexane (f) Tert-Butyl alcohol
-
= You are designing a small cart and ramp system with a rubber band that has a spring constant k 140 N/m. The cart has a mass of m = 0.74 kg, and it is tied to the rubber band. The frictionless ramp...
-
Now the person moves his hand back and forth several times to produce several waves. You freeze the movie and get this snapshot. Underline your answer for each situation: 5. If you advance the movie...
-
An electron and a positron are moving toward each other and each has speed 0.490c in the lab frame. Part A What is the kinetic energy of each particle? Enter your answers in joules separated by a...

Study smarter with the SolutionInn App


