The molecules nitromethane and 2-propanol have roughly the same shape and molecular mass. Liquid 2-propanol contains hydrogen
Question:
The molecules nitromethane and 2-propanol have roughly the same shape and molecular mass.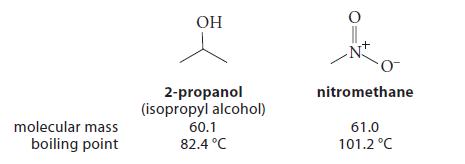
Liquid 2-propanol contains hydrogen bonds, but liquid nitromethane does not. Yet nitromethane has the higher boiling point. Why does nitromethane have such a high boiling point? What physical properties of the two molecules could you look up to support your answer?
Transcribed Image Text:
molecular mass boiling point OH 2-propanol (isopropyl alcohol) 60.1 82.4 °C nitromethane 61.0 101.2 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
nitromethane and 2propanol have similar molecular mass but different boiling points Nitromethane has ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Discussion 1- One key component of descriptive writing is showing the reader something rather than telling the reader. For example, I could tell you that I was cold. Or, I could show you: I shivered,...
-
In order to evaluate lim f(a+h)-f(), it is necessary to evaluate f(a + h). h xa For f(x) = x 3, f(a+h) =
-
Was there sufficient evidence to prove that Reyes violated the AECA and related regulations?
-
Lois and Kam are married and file a joint return. Lois earns $64,500 and Kam earns $30,000. Their adjusted gross income is $103,000. Determine the maximum IRA contribution and deduction in each of...
-
Refer to the information in Exercise 16-12. Prepare a process cost summary using the FIFO method. (Round cost per equivalent unit calculations to two decimal places.) Data From Exercise 16-12 The...
-
Bertha's Bathworks produces hair and bath products. Its biggest customer is a national retail chain that specializes in such products. Bertha Jackson, the owner of Bertha's Bathworks, would like to...
-
Provide an objective setting paragraph on HP Inc.'s decision to increase the R&D investment to use towards their partnership with Poly Solutions to enhance Microsoft Teams flexibility
-
(a) One of the following compounds is an unusual example of a salt that is soluble in hydrocarbon solvents. Which one is it? Explain your choice. (b) Which of the following would be present in...
-
Without consulting tables, arrange the compounds within each of the following sets in order of increasing boiling point, and give your reasoning. (a) 1-hexanol, 2-pentanol, tert-butyl alcohol (b)...
-
The accounts receivable balance for Renue Spa at December 31, 2017, was $61,000. Also on that date, the balance in the Allowance for Doubtful Accounts was $3,750. During 2018, $2,100 of accounts...
-
1.What fear tactics can we use to help people reduce plastic waste? 2. How can brands change their behavior in order to encourage their customers to use eco-friendly products? 3. What baby steps can...
-
Calculate the impulse from the below applied force versus time graph. () F(newtons) 3 2 2 4 6 8 - 1 (sec ) 10 8 N.S 13 N.S 11 N.S 10 N.S ()
-
You just purchased a share of SPCC for $101. You expect to receive a dividend of $5 in one year. If you expect the price after the dividend is paid to be $114, what total return will you have earned...
-
Analysis of the Industry-Activision Blizzard Strategic Group(s) in which the company exists and competitors in it/them. Intensity of rivalry among existing competitors (Use Porters 5 Forces for...
-
How Changes in Economic Conditions Could Potentially Affect the Funds and policies that direct the use of these funds. A change in the demand or need for the PMHC program or its services could affect...
-
What is meant by investment in human capital? Use this concept to explain (a) Wage differentials (b) The long-run rise of real wage rates in the United States.
-
Express mass density in kg/m3 and weight density in lb/ft3. 1. Find the mass density of a chunk of rock of mass 215 g that displaces a volume of 75.0 cm3 of water. 2. A block of wood is 55.9 in. x...
-
Identify the most acidic site in thesecompounds: NH2 CH.COCH,CH,CH3 c) a) b) CH3 e) CH;CH,CH,COH d) CH,CH,CCH,CH3
-
Suggest explanations for the origins of "ibu," "pro," and "fen" in the name ibuprofen. Provide a systematic name for thiscompound OH O,N. NO2 NO2 Picric acid
-
The pK a for the picric acid is 0.42. Explain why it is such a strong acid.
-
Draw a graph of a country in autarky equilibrium using as few curves and points as possible. Assume that this country only produces 2 goods (you decide what they are) and there is increasing...
-
Analyze how market microstructure theory seeks to explain stock price formation and evaluate how it differs from the efficient market hypothesis.
-
If the exchange rate is one dollar to 10 South African rand, how many dollars does it take to buy an item that costs 400 rand?

Study smarter with the SolutionInn App


