The reaction given in Fig. P18.74 occurs readily at 95C (X = halogen). The relative rates of
Question:
The reaction given in Fig. P18.74 occurs readily at 95°C (X = halogen). The relative rates of the reaction for the various halogens are 290 (X = F), 1.4 (X = Cl), and 1.0 (X = Br). When a nitro group is in the para position of each benzene ring, the reaction is substantially accelerated. Give a detailed mechanism for this reaction, and explain how it is consistent with the experimental facts.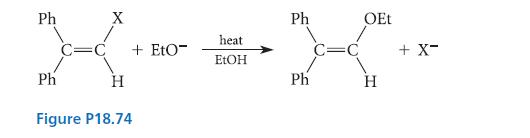
Transcribed Image Text:
Ph X X Ph H C=C + Eto- Figure P18.74 heat EtOH Ph Ph C=C OEt H + X-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Because vinylic halides do not undergo Sy2 or Syl reactions this s...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
This data table contains the listed prices and weights of the diamonds in 48 rings offered for sale in The Singapore Times. The prices are in Singapore dollars, with the weights in carats. Use price...
-
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P27.51. The equilibrium mixture contained compound A (22%), a single stereoisomer of B...
-
This unit addresses the role of the technology professional in an organization and how information technology (IT) systems influence organizational strategies. This case study analyzes two retailers'...
-
Write a filter that reads text from an input stream and prints it to an output stream, removing any lines that consist only of whitespace.
-
A light beam moves along the y axis with speed c in frame S, which is moving to the right with speed V relative to frame S. (a) Find the x and y components of the velocity of the light beam in frame...
-
Janet Boyle intends to deposit $300 per year in a credit union for the next 10 years, and the credit union pays an annual interest rate of 8%. a. Determine the future value that Janet will have at...
-
Draw a UML sequence diagram for a use case for an attack on a car through its telematics unit. The attack first modifies the software on the telematics unit, then modifies software on the brake unit....
-
On June 1, 2016, Hamiota Inc. (Hamiota) issued a $250,000,000 bond with a 5 percent coupon rate and a maturity date of May 31, 2026. Interest will be paid annually on May 31. Hamiota's year-end is...
-
On July 31, 2022, Edgmont Co. purchased $ 44 comma 000 of inventory on a one-year, 9-percent note payable. Interest payable on December 31, 2022, was $1 comma 650. Show what Edgmont Co. reports for...
-
Give the structure for each of the following compounds. (a) Isobutryaldehyde (b) Valerophenone (c) O-bromoacetophenone (d) G-chlorobutyraldehyde (e) 3-hydroxy-2-butanone (f)...
-
Predict the product of the Stille reaction between ethynyltrimethylstannane, HC CSn(CH 3 ) 3 , and phenyl triflate, PhOTf, in the presence of Pd(PPh 3 ) 4 and excess LiCl.
-
Which characteristic of a corporation is a disadvantage? a. Mutual agency b. Double taxation c. Limited liability d. None are disadvantages.
-
For each of the following situations, explain how current consumption will change according to the random walk hypothesis: a) The government increases taxes to close the budget deficit, but the size...
-
A government committed to long-run fiscal discipline (i.e., low or zero budget deficits) usually conducts contractionary fiscal policy at some point to reduce the government deficit. If that action...
-
A Singapore company has a subsidiary in England and another subsidiary in the United States. Both subsidiaries maintain their books and accounting records in their respective currencies. The...
-
Co B is the issuer of a tranche of mandatorily redeemable convertible preference shares (MRCPS) that was issued on the following terms: Required 1. Identify the elements included in the MRCPS. 2....
-
Scenario: P Co decreases ownership interest in X Co from 80% to 50% Required Determine the amounts of the following items (if any) arising at the date of the most recent transaction in each scenario:...
-
George Eastman was born in 1854 in Upstate New York. A high school dropout because of the untimely death of his father and the ensuing financial needs of his family, Eastman began his business career...
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
N-Methylpyrrolidine has a boiling point of 81 °C, and piperidine has a boiling point of 106 °C. (a) Explain this large difference (25 °C) in boiling point for these two isomers. (b)...
-
Predict the hybridization, geometry, and bond angles for the carbon and nitrogen atoms in acetonitrile (CH3C N:).
-
Which of the following pure compounds can form hydrogen bonds? Which can form hydrogen bonds with water? Which ones do you expect to be soluble in water? (a) (CH3CH2)2NH (b) (CH3CH2)3N (c)...
-
Figure 2 shows a rectangular sheet of metal which is 120 cm long and 90 cm wide. A square of side x cm is cut from two corners as shown in the diagram and the sides are then folded up to make a tray...
-
What are the effects of organizational justice perceptions on employee trust and cooperation? How do psychological contracts influence employee expectations and organizational obligations?
-
The ratio of Jake's compact discs to those of Andrea is 3:2. When they each had two fewer discs it was 5:3. How many discs does Jake have now?

Study smarter with the SolutionInn App


