The structure of cyanocobalamin, one of the forms of vitamin B12. Notice that cyanocobalamin is a complex
Question:
The structure of cyanocobalamin, one of the forms of vitamin B12. Notice that cyanocobalamin is a complex of the transition metal cobalt (Co).
Characterize this compound in the following ways:
(a) The oxidation state of the cobalt
(b) The dn count (that is, the value of n)
(c) The total electron count around the metal
Figure P18.65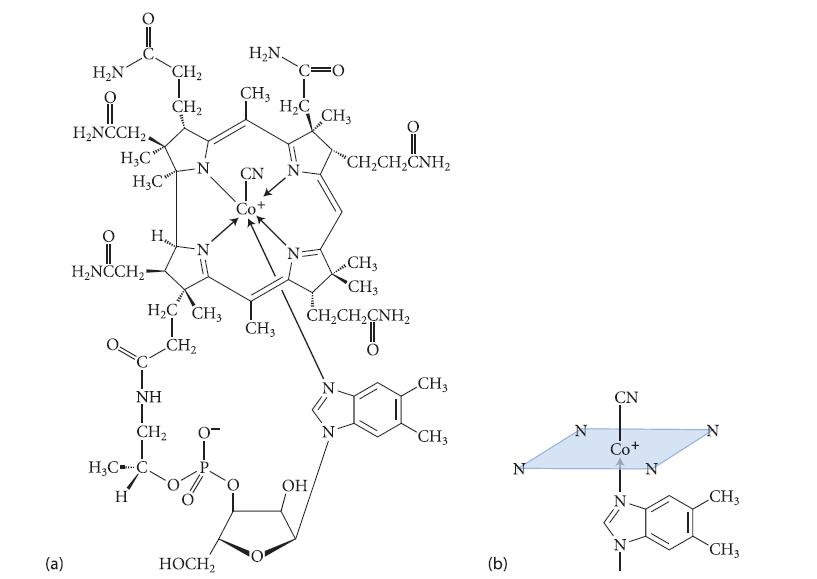
Transcribed Image Text:
(a) H₂N i H₂NCCH₂. H3C" H₂C H i H₂NCCH₂- NH CH₂ CH₂ H₂C CH3 T CH₂ T -N CH₂ 0- T H₂CC. H HOCH2 H₂N. CH3 CN Co+ CH3 H₂C N: CH3 OH, K *CH,CH,CNH, CH3 CH3 CH,CH,CNH, N PRAESE CH3 CH3 (b) N Z CN Co+ CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a The oxidation state is the number of valence electrons in the neutral ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
QUESTION 21 Which of the following is not a wrapper class? A. String B. Integer C. Character D. Double QUESTION 22 The conversion of an object of a wrapper class to a value of its associated...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A transition metal X forms an oxide of formula X2O3. It is found that only 50% of X atoms in this compound are in the +3 oxidation state. The only other stable oxidation states of X are +2 and +5....
-
A chemical factory discharges waste products into a river. The waste products affect a fishery downstream. Suppose MD = 6E and MAC = 600 - 4E. Consider a liability law requiring the polluter to...
-
Northwest Magazine sells subscriptions for $36 for 12 issues. The company collects cash in advance and then mails out the magazines to subscribers each month. Requirement 1. Apply the revenue...
-
A family is relocating from St. Louis, Missouri, to California. Due to an increasing inventory of houses in St. Louis, it is taking longer than before to sell a house. The wife is concerned and wants...
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
Ashley Williams opened Ashleys Maids Cleaning Service on July 1, 2017. During July, the company completed the following transactions. July 1 Invested $14,000 cash in the business. 1 Purchased a used...
-
The figure depicts Jack - in - the - box: "Jack" is attached inside a box by a spring, as shown. You estimate Jack's mass to be 0 . 4 k g . As so often happens, a sign tells you the spring constant:...
-
Complete each reaction given in Fig. P18.70, by giving the major organic product(s), and explain your reasoning. No reaction may be an appropriate response. (a) (b) (c) (d) ON- ON. Cl NO CH3CH- -F +...
-
A mixture of p-cresol (4-methylphenol), pK a = 10.2, and 2,4-dinitrophenol, pKa = 4.11, is dissolved in ether. The ether solution is then vigorously shaken with one of the following aqueous...
-
Hamilton, a U.S. corporation, reports the following results from its current year activities: U.S.-source taxable income . . . . . . . . . . . . . . . . . . . . . . . . . . . $1,000,000...
-
Human Resources, whether or not it is an area to enjoy as. a career. If yes, what particular facet of HR appeals? how would t it would impact the operations of the field of business management?
-
Find a politically oriented website and analyze the material using George's six questions for evaluating sources (George, 2008). What does your analysis say about the material on the website?
-
Referring to the Altex Corporation case study, why was a risk management plan considered unnecessary? Referring to the Altex Corporation case study, How effective will the risk management plan be if...
-
I am a new employee in your marketing department and have never heard of CRM. Educate me by discussing what CRM is as both a noun and a verb, what we use CRM tools for, what the benefits of using a...
-
The Intercultural Briefing * helps them strengthen the workplace culture in New Zealand. Realizing that they must work with Filipinos and Germans across cultures, they want to make suggestions on how...
-
Refer to Exhibit, which portrays the three types of allocation procedures used in two-stage allocation. Give an example of each of these allocation procedures in a hospital setting. The ultimate cost...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
(a) Write resonance structures for the phthalimide anion that account for the acidity of phthalimide. (b) Would you expect phthalimide to be more or less acidic than benzamide? Why? (c) In step 3 of...
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) Product of (c) heat (e) (f) (g) Aniline + propanoyl chloride (h) Tetraethylammonium hydroxide heat (i) p-Toluidine...
-
Starting with benzene or toluene, outline a synthesis of each of the following compounds using diazonium salts as intermediates. (You need not repeat syntheses carried out in earlier parts of this...
-
operations: Create a star topology network and perform the following a) Use a router for the network and apply DHCP setting on it such that a range that includes 5 IP addresses are exempted for...
-
1.2 Imagine you've been hired as an IT consultant for a small business that is expanding its operations and needs to set up a new network to accommodate its growing number of employees and devices....
-
You were recently employed as a Network Engineer at UCT in the ICTS department and received a complaint from a student transferring data between two sites directly connected to the UCT network saying...

Study smarter with the SolutionInn App


