Using abbreviated structures like the ones in Eq. 25.9a, give a curved-arrow mechanism for the reduction of
Question:
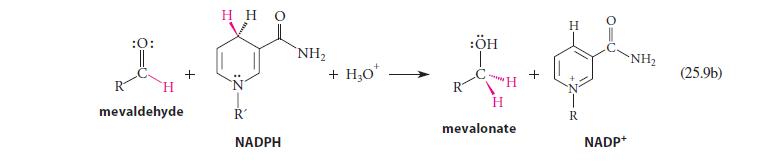
Using abbreviated structures like the ones in Eq. 25.9a, give a curved-arrow mechanism for the reduction of mevaldehyde shown in Eq. 25.9b.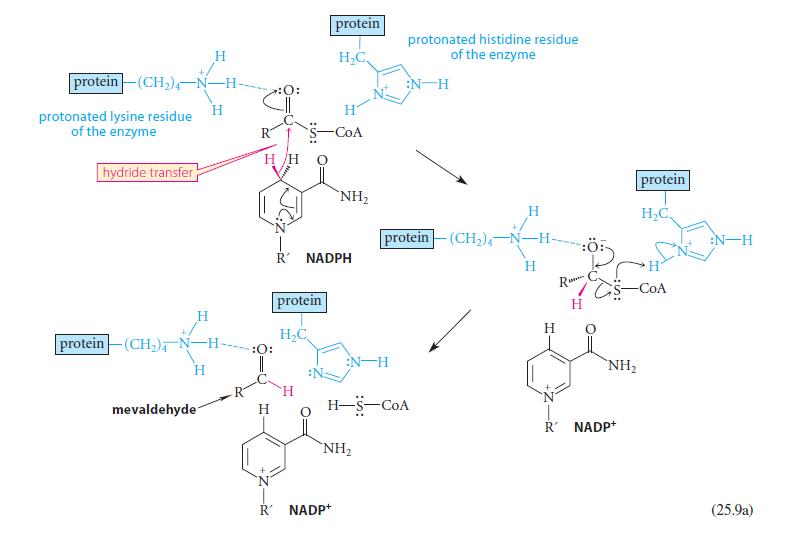
Transcribed Image Text:
protein (CH₂)-N-H- protonated lysine residue of the enzyme hydride transfer H mevaldehyde H H protein (CH₂), N-H- H H/H :O: :O: H protein H₂C H protein H₂C, R NADPH -CoA NH₂ R' NADP+ NH₂ protonated histidine residue of the enzyme :N H protein(CH₂)-N- -H H-S-COA H H H R TG H NH₂ R NADP+ protein H₂C -COA :N-H (25.9a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Despite the central phosphorus being tetrahedral with four different gro...View the full answer

Answered By

Monica Malik
I have been teaching Physics and Mathematics for over five years now. I ran a tutorial in Goa where I tutored students between the age of five and eighteen. Earlier this year (2019) I got TEFL certification and now I teach English to students from six countries for two different online portals.
I volunteered in India, Nepal, Srilanka, Thailand and Cambodia and enjoy teaching a lot. I am looking forward to connecting with more students so the learning and teaching can continue.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using abbreviated structures like the ones used in this section, outline the steps that convert hexanoyl-ACP into octanoyl-ACP during fatty-acid biosynthesis.
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The hydrolysis of phosphotyrosine esters in proteins is catalyzed by a family of enzymes called protein phosphotyrosine phosphatases. These hydrolyses in many cases involve phosphoenzyme...
-
John dies on 3 March 2021. Between 6 April 2020 and 3 March 2021, he has capital gains of 1,200 and capital losses of 15,400. His net gains in recent tax years (and the annual exemption for each...
-
Using Table 40-1, find the Q values for the following reactions: (a) 1 H + 3 H 3 He + n + Q and (b) 2 H + 2 H 3 He + n + Q Table 40-1 Atomic Masses of the Neutron and Selected Isotopes Element...
-
The board of Wicker Home Health Care, Inc., is exploring ways to expand the number of shares outstanding in an effort to reduce the market price per share to a level that the firm considers more...
-
Let \(d S_{t}=S_{t^{-}}\left(b d t+\sigma d W_{t}+\phi d M_{t} ight)\) where \(b, \sigma\) and \(\phi\) are constant coefficients and \(\phi>-1\). Let \(Y_{t}=\left(S_{t} ight)^{-1}\). Prove that \[d...
-
The following independent cases describe facts concerning the ownership of racing bicycles. (a) Piero Niccolo, winner of the 2009 MilanSan Remo cycling classic, purchased a new Fierro bicycle for...
-
Darren applies a mark-up on cost of 40% to his purchases. (a) How much will his gross profit be if Darren's sales for the year are 98,000? (b) What would the cost of sales be if the mark-up is 25%?
-
The following chiral phosphate ester cannot be isolated in optically active form. Explain. O CH3O-P-OH OCHCH3
-
The side chain R of the amino acid serine is CH 2 OH. Draw the structure of the phosphate monoester of serine.
-
1. What characteristics would you expect to have in a robot that provides emotional support to patients? 2. Can you think of other applications where robots such as the Huggable can play a helpful...
-
Around $500 billion in U.S. Treasuries are traded each day and many countries hold U.S. Treasuries, as they are considered very safe. For example, as of May 2022, Japan held $1,213 billion in U.S....
-
You read two articles online. The first says GDP grew by 5% last quarter, but the second states that the economy grew by 3.4% over the same time period. What do you think explains the difference in...
-
How do businesses change their prices at different levels of output, and how does this lead to an upward-sloping aggregate supply curve?
-
Seana owns a small pet shop and expects inflation to be 3% next year. By how much does Seana expect her marginal costs to change? By how much does she expect her competitors prices to change?
-
In Uganda, GDP per person is $2,280 per year, and in Japan it is $42,338. What do you think is likely to happen to each countrys GDP per person if they both increase their physical capital per person...
-
Suppose the average checkout tab at a large supermarket is $65.12, with a standard deviation of $21.45. Twenty-three percent of the time when a random sample of 45 customer tabs is examined, the...
-
(a) Find the equation of the tangent line to f(x) = x 3 at the point where x = 2. (b) Graph the tangent line and the function on the same axes. If the tangent line is used to estimate values of the...
-
Outline a Preparation of the following compounds from aniline and any other reagents. Sulfathiazole, a sulfa drug. H2N sulfathiazole
-
Outline a synthesis for each of the following compounds from the indicated starting materi-als using a reaction sequence involving a diazonium salt. (a) 2-bromobenzoic acid from o-toluidine...
-
What two compounds would react in a diazo coupling reaction to form FD & C Yellow No. 6?
-
How does the integration of socio-ecological resilience perspectives, such as the adaptive cycle or paparchy theory, elucidate the dynamic interplay between individuals, communities, and ecosystems...
-
20. What is the output of the following C++ code? (4) a. vector classList; classList.push_back("Sheila"); classList.push_back("Anita"); classList.push_back("Cecelia"); classList.push_back("Henry");...
-
A shopkeeper mixes peanuts(cost per kg = 25) and walnuts(cost per kg = 500) to make a 20 kg mixture, which costs 4300 rupees. How many kg of peanuts and walnuts each are put in the mixture?

Study smarter with the SolutionInn App


