What alkyl halide and what alkene would yield each of the following cyclopropane derivatives in the presence
Question:
What alkyl halide and what alkene would yield each of the following cyclopropane derivatives in the presence of a strong base?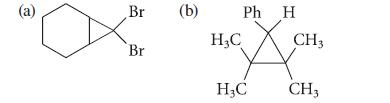
Transcribed Image Text:
(a) Br Br (b) Ph H Ă H₂C H₂C CH 3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a cyclohexene Br...View the full answer

Answered By

Aadarsh Jain
I am a B.COM Graduate from IGNOU. Since 2018 I have been teaching Accounting & Taxation to B.COM Students. Teaching is a noble profession. One does not go into teaching for the money but rather for the twinkle in the eyes of a student when they understood the concept i taught them or figured out for themselves how to solve a problem based on guidance. Once i find my students not able to understand a particular concept i always ready to adopt new methods of teaching to explain to them. While teaching, I feel good and happy when my students feel also good and happy because they learnt the key points of my lectures.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From which alkene could each of the following cyclopropane derivatives be prepared using the Simmons- Smith reaction? CH3
-
Starting with an appropriate alkyl halide and base, outline syntheses that would yield each of the following alkenes as the major (or only) product: (a) (b) (c) (d) (e)
-
For each of the following descriptions draw the structure of a compound that fits the description. (Note: There are many correct answers for each of these problems.) a) An alkyl halide that produces...
-
The following information has been extracted from the financial statements of XYZ Ltd for the year ended 31 December 2022: Income Statement Sales revenue: $3,000,000 Cost of goods sold: $1,200,000...
-
The General Fund trial balance of the City of Cordes as of January 1, 2012, was as follows: The following data pertain to General Fund operations for the City of Cordes for the fiscal year ended...
-
On November 28, 1990, Federal Reserve Chairman Alan Greenspan told the House Banking Committee that despite possible benefits to the U.S. trade balance, ''a weaker dollar also is a cause for...
-
Using only the factor formulas given in Table 2.6, derive Equation 7.6 starting with Equation 7.3. TABLE 2.6 Summary of Discrete Compounding Interest Factors. To Find Given Factor Symbol Name P F...
-
The Wallace Stationary Company purchases paper from the Seaboard Paper Company. Wallace produces stationary that require 1,415,000 sq. yards of stationary per year. The cost per order for the company...
-
The KLM Partnership, which uses the accrual method of accounting, is owned equally by Karen ( cash method taxpayer ) , and LM corporation ( accrual method taxpayer ) . Karen is a real estate...
-
(a) Give the structures of two isomeric alkylmagnesium bromides that would react with water to give propane. (b) What compounds would be formed from the reactions of the reagents in (a) with D 2 O?
-
Give the products of the following reactions. Show the curved-arrow notation for each. (a) HC-Li + CHOH (b) (CH,),CHCH, MgCl + H,O
-
Implement the linear optimization model developed for Leisure Furniture in Problem 29 in a spreadsheet and use Excel Solver (see Supplement D) to determine the optimal production quantities of each...
-
Has the theory of purposeful work behaviour any significance when it comes to integrating personality and motivation theories.
-
Suppose the interest rate is 6.8% APR with monthly compounding. What is the present value of an annuity that pays $108 every six months for six years?
-
What is the significance of stereotyping in person perception?
-
You can earn $38 in interest on a $1000 deposit for eight months. If the EAR is the same regardless of the length of the investment, determine how much interest will you earn on a $1000 deposit for...
-
You have just purchased a home and taken out a $460,000 mortgage. The mortgage has a 30-year term with monthly payments and an APR of 6.08%. a. How much will you pay in interest, and how much will...
-
MicroTech Corporation is subject to a 35 percent income tax rate. Given the following information about the firms capital structure, calculate the corporations weighted-average cost of capital(WACC):...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
How would you carry out the following conversions? More than one step is needed in eachcase. "
-
Synthesize the following compounds using 1-butyne as the only source of carbon, along with any inorganic reagents you need. More than one step may be needed. (a) 1, 1, 2, 2-Tetrachlorobutane (b) 1, 1...
-
How would you synthesize the following compounds from acetylene and any alkyl halides with four or fewer carbons? More than one step may herequired. (a) CH3CH2CH2C=CH (b) H2C%3CCH CH CH CHH2H3D (d)...
-
Perkins, Inc. had net sales of $1,530,000 during the year. On January 1, Perkins' accounts receivable was $320,000. On December 31, Perkins' accounts receivable was $400,000. What was Perkins'...
-
How Having effective audit and information disclosure can solve most corporate governance problems. Discuss
-
The Extan division of World Corporation had $9 million of total assets, $3 million of current liabilities and a profit of $1 million. The company defines invested capital as total assets. Calculate...

Study smarter with the SolutionInn App


