Which of the following molecules is likely to be planar and which nonplanar? Explain. :O: oxepin tropylium
Question:
Which of the following molecules is likely to be planar and which nonplanar? Explain.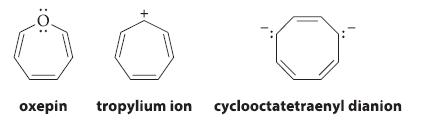
Transcribed Image Text:
:O: oxepin tropylium ion cyclooctatetraenyl dianion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The species with 4n 2 electrons are likely to be plan...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
There is no trade-off between customer-response time and on-time performance. Do you agree? Explain
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
From a list of 468 small 2-year colleges a simple random sample of 100 colleges was drawn. The sample contained 54 public and 46 private colleges. Data for number of students (y) and number of...
-
Watch the sample Persuasive Presentation Video link given in this module on Success is a Continuous Journey by Richard St. John .Answer the following questions in detail and use complete sentences....
-
Venture Company earned $8,000 of service revenue on account during 2010. The company collected $5,200 cash from accounts receivable during 2010. Required Based on this information alone, determine...
-
Suppose in the previous problem that HISC always needs a conveyor belt system; when one wears out, it must be replaced. Which system should the firm choose now? In the previous problem, Hagar...
-
In February 2007, The Elliot Group, Inc., an Illinois real estate developer, made a deal with the Village of Arlington Heights to develop property in that village. Arlington Market, LLC, was...
-
Takemoto Corporation borrowed $60,000 on November 1, 2012, by signing a $61,350, 3-month, zero-interest-bearing note. Prepare Takemotos November 1, 2012, entry; the December 31, 2012, annual...
-
The interest rate is fixed at 10%. You are offered an asset that pays $1,000 from now until eternity at yearly intervals. You find its present value by calculating the sum of the discounted annual...
-
A colleague, Ima Hack, has subjected isoprene to catalytic hydrogenation to give isopentane. Hack has inadvertently stopped the hydrogenation prematurely and wants to know how much unreacted isoprene...
-
Give the principal product(s) expected, if any, when trans-1,3-pentadiene reacts under the following conditions. Assume one equivalent of each reagent reacts unless noted otherwise. (a) Br 2 (dark)...
-
Determine whether the given function is a solution of the differential equation. y = tan x; y' y 2 = 1
-
As the price of a service rises, _______. a) the consumer surplus decreases b) the consumer surplus increases c) the consumer surplus may increase or decrease
-
Variable costs change with _____.
-
A products utility to a buyer is measured by ______. a) its usefulness b) its price c) how much the buyer is willing to pay for it d) none of the above
-
When demand rises and supply stays the same, ______. a) equilibrium quantity rises b) equilibrium quantity declines c) equilibrium quantity stays the same.
-
If a service is free, you will consume more and more of it until ______. a) your marginal utility is zero b) your total utility is zero c) both your marginal utility and your total utility are zero...
-
Hersh inherits $50,000 from his grandfather. He receives the money on January 1 and immediately invests $25,000 in General Motors bonds that pay 8% annual interest and $25,000 in Lane County highway...
-
When the concentration of a strong acid is not substantially higher than 1.0 10-7 M, the ionization of water must be taken into account in the calculation of the solution's pH. (a) Derive an...
-
Halogenated compounds arc particularly easy to identify by their mass spectra because both chlorine and bromine occur naturally as mixtures of two abundant isotopes. Chlorine occurs as 35C1 (75.8%)...
-
By knowing the natural abundances of minor isotopes, its possible to calculate the relative heights of M+ arid M + 1 peaks. If 13C has a natural abundance of 1.10%, what are the relative heights of...
-
Propose structures for compounds that fit the following data: (a) A ketone with M = 86 and fragments at m/z = 71 and m/z = 43 (b) An alcohol with M + = 88 and fragments at m/z = 73, m/z = 70, and m/z...
-
Why must raw data, such as that compiled by the GFOA, be converted to useful geographical and population strata benchmarks?
-
1. Give an example of each circle part using the diagram below. G H a) Center: b) Radius: c) Chord: d) Diameter: e) Secant: f) Tangent: g) Point of Tangency:. h) Minor Arc: 1) Major Arc: j)...
-
The Precision Widget Company had the following balances in their accounts at the end of the accounting period: Work in Process Inventory $45,000 Finished Goods Inventory $67,500 Cost of Goods Sold...

Study smarter with the SolutionInn App


