Which of the salts shown in Fig. P6.49 should have identical solubilities in methanol? Explain. Ph HC
Question:
Which of the salts shown in Fig. P6.49 should have identical solubilities in methanol? Explain.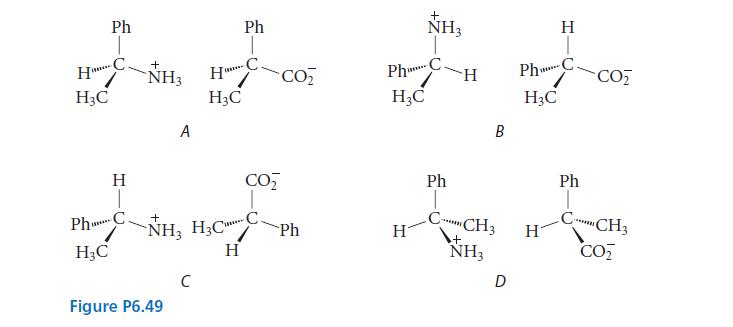
Transcribed Image Text:
Ph
HC
H.C
H
+
NH3
A
Figure P6.49
H;C
****
+
Ph C
Ph
HC
H.C
H
+
NH3
A
Figure P6.49
H;C
****
+
Ph C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Salts that are either identical or enantiomers should have identical solubilities The solution to th...View the full answer

Answered By

Emily Grace
With over a decade of experience providing top-notch study assistance to students globally, I am dedicated to ensuring their academic success. My passion is to deliver original, high-quality assignments with fast turnaround times, always striving to exceed their expectations.
4.90+
3+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain the order of water solubilities for the compounds in each of the following groups, (a) Ethanol > chloroethane > ethane; (b) Methanol > ethanol > 1-propanol.
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
Can you identify foreign jobs that are being performed inside the United States?
-
elow is selected financial information for SunRise Company. Selected Balance Sheet Data - As of Dec. 31, 2018 Dec. 31, 2017 Cash and short-term investments $ 958,245 $ 745,800 Accounts Receivable...
-
Morality clearly enters this case. In what way does Bieber argue that morality supports his claim?
-
ADC, Inc., is a corporation that was formerly a three-person partnership. It is in the business of acquiring software and distributing it to accounting firms that need specialized software in their...
-
While the electromagnetic interaction between the photons and the charged electrons and positrons kept them in equilibrium with each other during the early universe, show that the direct...
-
A number of graphs displaying cost behavior patterns are shown below. The vertical axis on each graph represents total cost, and the horizontal axis represents level of activity (volume). Required:...
-
Amazon Sales $177,866 Accounts receivable: Beginning of year 20,816 End of year 16,677 Determine the accounts receivable turnover. For interim computations, carry amounts out to one decimal place....
-
Draw the structures of the possible stereoisomers for the compound below, assuming in turn (a) Tetrahedral, (b) Square planar, (c) Pyramidal geometries at the carbon atom. For each of these...
-
(a) Explain why an optically inactive product is obtained when (2)-3-methyl-1-pentene undergoes catalytic hydrogenation. (b) What is the absolute configuration of (1)-3-methylhexane if catalytic...
-
Describe the basic accounting equation that provides the structure for the balance sheet. Define the three major components reported on the balance sheet.
-
Kane's business is producing a single product K. He collected the following information between production volume and maintenance costs, and now he is finding whether there is a linear relationship...
-
Reword the following Selling a property is one of the biggest financial transactions you'll make, at OBrien Real Estate we aim to take the stress out of the equation and deliver a result over and...
-
The budgeted variable MOH rate at is $5.20/machine hour, where every unit requires 3 machine hours. Additionally, the following fixed MOH costs are anticipated for the production: depreciation of...
-
A construction company sells a house with a FMV of $ 600 comma 000 to a shareholder of the corporation for $ 475 comma 000. The shareholder is liable for income tax on the $ 125 comma 000 difference....
-
If an individual shareholder of a corporation, who is in the business of lending money, receives a loan from the corporation with terms that are equivalent to that of clients, the principal amount...
-
What is a sunk cost? Provide an example of a sunk cost other than one from this book. Why are such costs irrelevant in making decisions about future actions?
-
What is the role of business risk analysis in the audit planning process?
-
Identify the carboxylic acid chloride that might be used in a Friedel-Crafts acylation reaction to prepare each of the followingacylbenzenes: (b) (a)
-
Write resonance structures for nitrobenzene to show the electron-withdrawing resonance effect of the nitro group.
-
Write resonance structures for chlorobenzene to show the electron-donating resonance effect of the chloro group.
-
1. What do you understand by variable in programming? [mark 1] 2. Explain what do you understand by the following line of code: [mark 1] int a, b, c; 3. What is the value of "sum" after the following...
-
Write the output in each of the following cases? for x in range(3): print(x) for y in [1,2,3]: print(y) for z in range(1,5): print(z) for pn in range(1,5,2): print(pn) for nn in range(1,-5,-2):...
-
For the given circuit diagram: (1) Obtain the Boolean expression step by step. (2) Obtain the truth table step by step. (3) From the result of (1) make the truth table of output F and compare with...

Study smarter with the SolutionInn App


