Which one of the following compounds could be prepared by the hydration of alkynes so that it
Question:
Which one of the following compounds could be prepared by the hydration of alkynes so that it is uncontaminated by constitutional isomers? Explain your answer.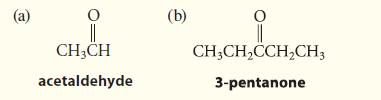
Transcribed Image Text:
(a) ||| CH3CH acetaldehyde (b) CH₂CH₂CCH₂CH3 3-pentanone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The compound that could be prepared by the hydration of alkynes without being contaminated by consti...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Indicate whether each of the following compounds could be prepared by a malonic ester synthesis. If so, outline a preparation from diethyl malonate and any other reagents. If not, explain why. (a)...
-
Indicate whether each of the following compounds could be prepared by a malonic ester synthesis. If so, outline a preparation from diethyl malonate and any other reagents. If not, explain why....
-
Differentiate between formal and behavioral roles, and describe how behavioral roles emerge during group interaction.
-
In a cash basis system, firms recognize revenues when they receive cash from customers and recognize expenses as they pay cash for goods and services. How does a cash basis system violate the...
-
Hinali Corporation issued $1,000,000 of 7 percent bonds on October 1, 2010 at 96. The bonds are dated October 1 and pay interest semiannually. The market interest rate is 8 percent, and Hinalas...
-
Two springs have spring constants \(k_{1}\) and \(k_{2}>k_{1}\). Connected as shown in Figure P8.53, they act like one spring. Compute the spring constant of the combination. Is the combination...
-
Aaron Heath is seeking part-time employment while he attends school. He is considering purchasing technical equipment that will enable him to start a small training services company that will offer...
-
Some time ago (1987) Professor Shaun Tyson produced the following prediction: Personnel work can be seen to be subject to conflicting and powerful pressures which are leading to the balkanization of...
-
An initiation step in the free-radical co-polymerization of styrene and 1,3-butadiene is the free-radical addition of a peroxidederived radical to a double bond of 1,3-butadiene: (a) Use the fishhook...
-
A compound X with the molecular formula C 5 H 10 O 2 has an IR spectrum with strong absorption in the 10001100 cm 1 region; very strong, broad absorption in the 30003600 cm 1 region; and no...
-
Which of the following is not a cost advantage independent of scale? A. proprietary technology B. favorable locations C. experience in the industry D. high volume of production
-
What are the three essential components of a mission?
-
Why might a person or unit in charge of strategic planning also be assigned responsibility for public relations, communications, and business development?
-
What is the purpose of a vision?
-
What are the advantages of using project charters? What might be some disadvantages?
-
How might an organizations culture reflect its established values?
-
Because of uncertainty in real estate markets, many homeowners are considering remodeling and constructing additions rather than selling. Probably the most expensive room in the house to remodel is...
-
Consider model (9.18). What is the effect on the model parameter estimates, their standard errors, and the goodness-of-fit statistics when (a) The times at risk are doubled, but the numbers of deaths...
-
Within each set, identify the ether that would not readily cleave with concentrated HBr and heat, and explain. Then give the products of ether cleavage and the mechanisms of their formation for the...
-
What reactants would be required to form the following compound by the Stille reaction? O2N OCH,CH
-
Give the product(s) expected (if any) when m-cresol or other compound indicated is subjected to each of the following conditions. (a) Concentrated H2SO4 (b) Br2 in CCI4 (dark) (c) Br, (excess) in...
-
Discuss the legal and ethical issues connected with border security and enforcement of the immigration laws.?
-
How does the state law conflict with federal immigration law? Support your response.
-
A stock Q is expected to pay a dividend of $6.20 annually in perpetuity. The stock has a beta of 1.20, the risk-free rate is 4.0%, and the market risk premium is 7.5%. 4 pts a What would you expect...

Study smarter with the SolutionInn App


