Work Study Problem 3.5 for the reverse of each reaction 3.16ac. CH3 H3C-C Br: I CH3 A
Question:
Work Study Problem 3.5 for the reverse of each reaction 3.16a–c.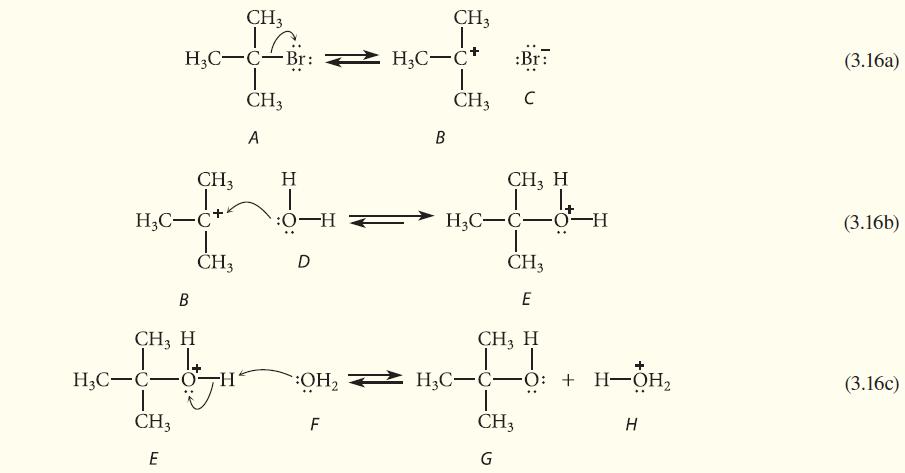
Transcribed Image Text:
CH3 H3C-C Br: I CH3 A CH3 H₂C-C+ B CH, H CH3 H3C-C-O -H I CH3 E H :0-H D F CH3 H₂C-C+ B T CH3 :Br C CH, H H3C-C-0-H CH3 E CH, H I OH₂ H3C-C-O: + H3C-CO: + H-OH₂ | CH3 G H (3.16a) (3.16b) (3.16c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a The reverse of reaction 316a is a Lewis acidbase association reaction ther...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-4. Ivan and Irene paid the following in 2012 (all by check or can otherwise be...
-
An autoglass company located in Detroit produces mirrors for automobiles. The company supplies auto mirrors to a nearby Ford (The Ford Motor Company, Dearborn, MI) plant. The demand for mirrors from...
-
Assume that the Tarasoff case is precedent. (In fact, many states have followed the ruling.) Make an argument that Ingram is liable.
-
Worrix Corporation manufactures and sells each year 3,000 premium-quality multimedia projectors at $12,000 per unit. At the current production level, the firms manufacturing costs include variable...
-
Describe the six categories of early warning signs of potential fraud.
-
Clapton Company manufactures custom guitars in a wide variety of styles. The following incomplete ledger accounts refer to transactions that are summarized for May: In addition, the following...
-
5. Suppose that acts on density operators for Q with Kraus representation () - k=1 Let U M, (C), and define operators = 1, k BkUkjAj. , Show that B,..., B., are also Kraus operators for E. 6. Even...
-
What is the pK a of an acid that has each of the following dissociation constants? (a) 10-3 (b) 5.8 10-6 x (c) 50
-
Write a Brnsted acidbase reaction in which act as conjugate acidbase pairs. H/-:H and CH,H/CH,:-
-
Bubbas Western Wear is a western hat retailer in Lubbock, Texas. Although Bubbas carries numerous styles of western hats, each hat has approximately the same price and purchase cost, as shown in the...
-
Question 2: Conversion Strategies Using Blackboard Ultra as an example, explain the following What conversion strategy was used to deploy Blackboard Ultra, the new version of Blackboard? What are the...
-
In a normal heart, which statement is true regarding the systemic and pulmonary circuits? A. Pressures are the same min both circuits B. Flow rate is the same in both circuits C. Velocity is the same...
-
Why can't the fire protection problems in a community be solved solely by building more fire stations and hiring more emergency response personnel? With fire sprinklers rated as 96% effective in...
-
21. A discrete-time signal x[n] is periodic with period 8. One period of its DFT harmonic function is {X[0],,X[7]}={3,4+ j5,-4-j3,1+ j5,-4,1- j5,-4+ j3,4-j5}. (a) What is the average value of x[n]?...
-
Discuss the role of taxonomic research in informing conservation policy and management strategies, including the identification of flagship species, the delineation of evolutionarily significant...
-
Suppose that when the interest rate on loans is 16 percent, businesses find it unprofitable to invest in machinery and equipment. However, when the interest rate is 14 percent, $5 billion worth of...
-
Experiment: Tossing four coins Event: Getting three heads Identify the sample space of the probability experiment and determine the number of outcomes in the event. Draw a tree diagram when...
-
The reaction of hydroxide ion with chloromethane to yield methanol and chloride ion is an example of a general reaction type called a nucleophilic sub- Situation reaction: HO- + CH3C1 CH3OH + C1-...
-
Ammonia reacts with acetyl chloride (CH3COCI) to give acetamide (CH3CONH2). Identify the bonds broken and formed in each step of the reaction, and draw curved arrows to represent the flow of...
-
The naturally occurring molecule ?-terpineol is biosynthesized by a route that includes the following step: (a) Propose a likely structure for the isomeric carbocation intermediate. (b) Show the...
-
Discuss the desiderata of effective Business Communication strategy Question Elucidate on the use of technology in the improvement of the communication process. Question Critically examine the...
-
The different NoSQL technologies were created to meet different needs. Choose one of the NoSQL data models and describe it. Explain what kind of applications would benefit from your chosen NoSQL data...
-
Learning Objectives At the end of this exercise, you should be able to: Learning Objective 1 . 5 LO 1 . 5 : Explain the value of studying management. Learning Objective 1 . 4 LO 1 . 4 : Describe the...

Study smarter with the SolutionInn App


