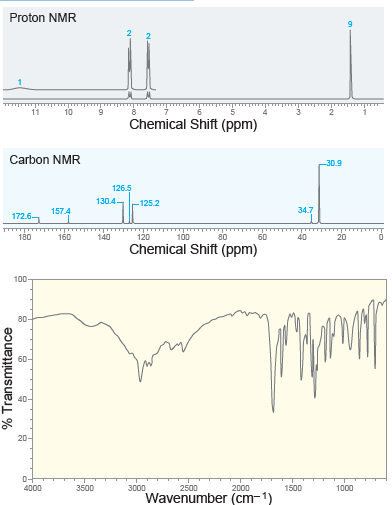
A compound with molecular formula C 11 H 14 O 2 exhibits the following spectra ( 1
Question:
Transcribed Image Text:
Proton NMR 11 Chemical Shift (ppm) Carbon NMR 30.9 126.5 130.4- -125.2 34.7 157.4 172.6 120 100 80 180 160 140 60 40 Chemical Shift (ppm) 100 80- По 60- 40- 20- 0- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-1) % Transmittance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When a compound with molecular formula C11H14O2 undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following 1H NMR spectrum. Identify the compound. 10 9 4
-
A compound with molecular formula C4H6O gives the infrared spectrum shown in Figure 13.34. Identify the compound. 3.5 4 5 12 13
-
Identify the following compounds. (Relative integrals are given from left to right across the spectrum.) a. The 1H NMR spectrum of a compound with molecular formula C4H10O2 has two singlets with an...
-
The passage indicates that the late 1850s Democrats: F. Were all Southern slaveholders who wanted to expand slavery into the territories. G. Used legislation in the early 1850s to support their...
-
What is breach of contract? What remedies are available to a party injured by a breach?
-
Evan is just leaving his house to visit his grandmother. Normally, the trip takes him 25 minutes on the freeway, going 55 mph. But tonight hes running 5 minutes late. How fast will he need to drive...
-
Consider a $4^{2} \times 3^{2} \times 2$ factorial design. a. How many factors are included in this design? b. How many levels are included in each factor? c. How many experimental conditions, or...
-
In Figure, a chain consisting of five links, each of mass 0.100 kg, is lifted vertically with constant acceleration of magnitude a = 2.50 m/s2. Find the magnitudes of (a) The force on link 1 from...
-
Back Country Airlines has the following accounts and balances as of their year-end, December 31st, 20X1 (assume all balances are "normal"). In good form (including proper headings subtotals, labels,...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
An ideal gas in a piston and cylinder assembly with adiabatic walls undergoes an expansion against a constant external pressure. Are S, S surroundings , and S total positive, negative, or zero?...
-
Is the equation valid for an ideal gas? Tf PV; -V;) T; Cy dT Lav = C, n2v, -v) %3D AS =
-
Rob is taking a learning test in which the time he takes to memorize items from a given list is recorded. Let M(t) be the number of items he can memorize in t minutes. His learning rate is found to...
-
Entity A prepares its financial statements on 31 July each year. The entitys balance sheet as at 31 July 2008 showed a liability for current tax of EUR 240,000. This was an estimate of the current...
-
What kinds of legal issues do you think stem cell research and genetic therapies will present in the coming years?
-
In todays economy, what are some examples of intrastate commerce? In other words, what business does not affect interstate commerce?
-
Given the information in Problem 28-17, how much more or less do the firms in this industry spend, in total, on the labor employed each hour as a consequence of establishment of the union wage W =...
-
An entity prepares financial statements to 31 December each year. The following events occurred-after 31 December 2009 but before the financial statements for the year ended 31 December 2009 were...
-
For what numbers is f(0) = cot not defined?
-
7. Baladna wants to analyze process that includes delivery by suppliers, production inside the company, transportation to to its customers and information systems. Then it also wants to find out...
-
What is the expected substitution product (including its stereochemical configuration) in the SN2 reaction of potassium iodide in acetone solvent with the following compound? (D = 2H = deuterium, an...
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
Prob. 1(a). Consider a stock currently trading at $40.25. For a strike price of $38, you want to price both a call and put option that matures 90 days from now. The volatility of the stock (2) is...
-
Wildhorse sells a snowboard, WhiteOut, that is popular with snowboard enthusiasts. Presented below is information relating to Wildhorse's purchases of WhiteOut snowboards during September. During the...
-
The following unadjusted trial balance is prepared at fiscal year-end for Nelson Company. Nelson Company uses a perpetual inventory system. It categorizes the following accounts as selling expenses:...

Study smarter with the SolutionInn App


