Arrange the following monomers in order of reactivity toward anionic polymerization. CI NO2
Question:
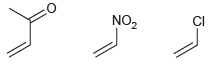
Transcribed Image Text:
CI NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
least r...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following monomers in order of reactivity toward cationic polymerization. NO2 CH3 OAc
-
Arrange each set of compounds in order of reactivity toward an E2 process: a. b. Br Br Br CI CI 'CI
-
Arrange each set of compounds in order of reactivity toward an E2 process: a. b. Br Br Br
-
Consider a post office with two employees and a single waiting line. On average, the inter-arrival time is 6 minutes. Standard deviation of inter-arrival time is 3.94 minutes. Average service time is...
-
June production generated the following activity in Bentley Chassis Company's Work-in Process Inventory account: June 1 balance .................................. $ 36,000 Direct materials used...
-
How would you influence the time of problem recognition for the following? a) Fire alarm battery replacement b) Gift basket c) Car tune-up d) Air conditioner filters e) Health insurance f) Vitamins
-
The following is the distribution of the hourly number of trucks arriving at a company's warehouse: Find the mean of this distribution, and using it (rounded to one decimal place) as the parameter...
-
The following questions illustrate nonannual compounding. a) One hundred dollars is placed in an account that pays 12 percent. How much will be in the account after one year if interest is compounded...
-
INDIVIDUAL DELIVERABLE A . While making your group decision, observe the members of the group and pay special attention to the group dynamics, conflicts that may or may not arise during the decision...
-
Barclay Brothers Company, the firm discussed in this module, thinks it underestimated the mean for its game Strategy. Rudy Barclay thinks expected sales may be 9,000 games. He also thinks that there...
-
Determine whether preparation of each of the following polymers would best be achieved via cationic addition or anionic addition. (a) (b) (c) (d) (e) (f) CN CN CN CN CN CN OMe OMe OMe OMe
-
Many monomers that readily undergo cationic polymerization, such as isobutylene, will not readily undergo anionic polymerization. Styrene, however, can be effectively polymerized via cationic,...
-
A gaseous fuel mixture that is 40 percent propane (C3H8) and 60 percent methane (CH4) by volume is mixed with the theoretical amount of dry air and burned in a steady-flow, constant pressure process...
-
How do resource utilization patterns and reallocation of resources relate to student performance?Items to consider in your discussion are why we reallocate resources and how do we reallocate so it...
-
Day Vision Inc. produces sunglasses. The company uses $1.28 in materials and $1.92 in labor to construct each pair. Over the course of one year, Day Vision incurs fixed costs of $580,000. Day Vision...
-
Select the sentence containing the correct subject-verb agreement. Each of the supervisors needs a planning calendar. Each of the supervisors need a planning calendar.
-
5. Let g be the function given by g(x) =x4 - 4x3 + 6x - 4x+ k, where k is a constant. A. On what intervals is g increasing? Justify your answer. g'(x) >0 for increasing function g(x) 4x-12x+12x-4 >0...
-
Clark's broker has suggested that she invest in bonds of one of the following companies: Issuer Price Windsor Ltd. $1,100 Edmonton Ltd. $1,500 Windsor's bonds were issued 9 years ago with a maturity...
-
Which of the following is/are usually included in an entrepreneurs business plan? a. Detailed description of the companys products and services. b. Discussion of the management team, including...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Show how you would synthesize the following derivatives from appropriate carbonyl compounds. (a) (b) (c) (d) (e) (f) CH,O OCH,
-
Predict the products formed when cyclohexanone reacts with the following reagents. (a) CH3NH2. H+ (b) Excess CH3OH. H+ (c) Hydroxylamine and weak acid (d) Ethylene glycol and p-toluenesulfonic acid...
-
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents. (a) PhMgBr, then H3O+ (b) Tollens reagent (c) Semicarbazide and weak acid (d) Excess ethanol and acid (e)...
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


