Assign a name for each of the following compounds. Be sure to assign the configuration of each
Question:
a.
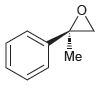
b.
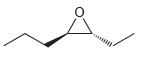
c.
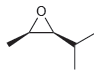
Transcribed Image Text:
Me
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a S2phenyl1 2epoxypropane or S1me...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Assign a name for each of the following compounds. a. b. c.
-
Assign a name for each of the following compounds. a. b. c.
-
Assign a name for each of the following compounds: (a) (b) (c) (d) (e) (f) NH
-
Use the accompanying graph of y = f(x). Does exist? If it does, what is it? lim f(x)
-
Cheney Company sold a 20-ton mechanical draw press for $67,700. The old draw press cost $90,300 and had a book value of $62,000. Prepare the journal entry to record the disposition.
-
Debate: Survey feedback can be a problematic OD technique because it permits people who are affected by organizational policies to generate data that speak against those policies.
-
The Arrow-Pratt relative risk aversion coefficient is \[\mu(x)=\frac{x U^{\prime \prime}(x)}{U^{\prime}(x)} .\] Show that the utility functions $U(x)=\ln x$ and $U(x)=\gamma x^{\gamma}$ have constant...
-
The inventory of Royal Decking consisted of five products. Information about the December 31, 2011, inventory is as follows: Disposal costs consist only of a sales commission equal to 10% of selling...
-
Selected transactions for Carla Vista Corporation during its first month in business are presented below. Sept. 1 Issued common stock in exchange for $ 2 1 , 8 0 0 cash received from investors. 5...
-
Consider the following two mutually exclusive projects. Whichever project you choose, if any, you require a 15 percent return on your investment. a. If you apply the payback criterion, which...
-
a. Using the relationships derived in Example Problem 7.1 and the values of the critical constants for water from Table 7.2, calculate values for the van der Waals parameters a, b, and R from z c , T...
-
Identify the reactants you would use to form a racemic mixture of each of the following epoxides: a. b. c. d. Mery Me
-
Consider either the purinosome or a multifunctional protein such as the CAD protein, and propose one or two experiments to determine whether the enzyme "channels" substrates through a multistep...
-
Consider the function Find the derivative of y with respect to . dy dx x - 3x+4
-
Calculate the theoretical maximum percentage efficiency of a coal fired power plant producing steam at 640C and rejecting into a low temperature sink at 20C.
-
Home Corp sold a machine for $50,000. Home Corp bought the machine for $55,000 several years ago and took $12,500 depreciation on the machine. What is the amount and character of Home Corp in profit...
-
Explore the application of nanomaterials and nanotechnology in the development of novel catalysts for sustainable chemical transformations, highlighting their enhanced activity, selectivity, and...
-
What would you do if the day after your presentation of your valuation report to the company, their stock holdings fell precipitously?
-
Assume that a person spins the pointer and is awarded the amount indicated by the pointer. Determine the persons expectation. $10 $5
-
Suppose that fraction used = / 1.0 + 0.1Mt. for some parameter 1. Write the discrete-time dynamical system and solve for the equilibrium. Sketch a graph of the equilibrium as a function of ....
-
Order the following monomers with respect to their expected reactivity toward anionic polymerization, and explain your answer: H2C = CHCH3, H2C = CHC N, H2C = CHC6H5
-
Polystyrene is produced commercially by reaction ol styrene with butyl lithium as an anionic initiator. Using resonance structures, explain how the chain-carrying intermediate is stabilized.
-
Vinylidene chloride, H2C = CCl2, does not polymerize in isotactic, syndiotactic, and atactic forms. Explain.
-
Sophia operates a large singing school with 22 employees. She receives advance fee payments from students in respect of each course (a course consists of 26 lessons). If a student does not attend all...
-
What role does agency play in the Hypodermic Model of Communication? Long after the model has been abandoned by experts in persuasion and mass media, why does this model live on as common sense?
-
Daniel is a resident individual who carries on a retail business but does not use the SBE method of accounting for income in his business. On 30 June of the current tax year he had trade debtors of...

Study smarter with the SolutionInn App


