Question: Below are two hypothetical compounds. a) Which compound would you expect to hold greater promise as a potential antihistamine? Explain your choice. b) Do you
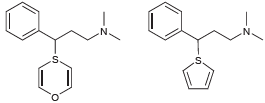
a) Which compound would you expect to hold greater promise as a potential antihistamine? Explain your choice.
b) Do you expect the compound you chose (in part a) to exhibit sedative properties? Explain your reasoning.
N.
Step by Step Solution
3.34 Rating (154 Votes )
There are 3 Steps involved in it
a The second compound holds greater promise as a potential antihistamine be... View full answer

Get step-by-step solutions from verified subject matter experts


