Classify each of the following compounds below as cis, trans, or not stereo-isomeric: , ,
Question:
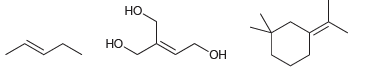
Transcribed Image Text:
но, Но но, НО Он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
trans HO H...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Classify each of the following compounds as an alkane, alkene, alkyne, alcohol, aldehyde, amine, and so forth. (a) (b) CH3-C¡CH (c) (d) (e) (f) Obtained from oil of cloves -2. Sex attractant...
-
Determine whether each of the following compounds is a cis isomer or a trans isomer a. b. c. d. e. f. Cl Br CH3 CH3 Br Br CH3 Cl CH3 CH3 CH
-
Draw both chair conformations for each of the following compounds. In each case, identify the more stable chair conformation: (a) Methylcyclohexane (b) Trans-1,2-Diisopropylcyclohexane (c)...
-
Evaluate the derivatives of the following functions. (z) = cot -1 z
-
Read the case study titled "Pre-Launch decisions which influence innovation success". Write a paper in which the following items are addressed. Discuss the necessity of short-term and long-term...
-
Criminal cases are assigned to judges randomly. The list of the criminal judges for Memphis, Tennessee (taken from the Tennessee Court System website, www.tsc.state.tn.us), is given in the table....
-
An orthotropic AS/3501 carbon/epoxy lamina (see Tables 2.2 and 4.1) is subjected to the plane stress condition \(\sigma_{x}=1000 \mathrm{MPa}, \sigma_{y}=50 \mathrm{MPa}\), and \(\tau_{x y}=50...
-
1. How do the innovation ideas described here support the creation and maintenance of a high-performance work system? What element(s) of a high-performance work system do these efforts relate to? 2....
-
S&L Financial buys and sells securities expecting to earn profits on short-term differences in price. Assume that on December 27. 2024, S&L purchased Coca-Cola bonds at par for $894,000 and sold the...
-
Listed below are nine technical accounting terms introduced in this chapter: Each of the following statements may (or may not) describe one of these technical terms. For each statement, indicate the...
-
Identify whether each of following pairs of compounds are enantiomers or diastereomers: a. b. c. d. e. f. OH OH .CI CI
-
For each of the following pairs of compounds, determine the relationship between the two compounds: a. b. c. d. e. f. g. h. i. j. k. l. Br Br
-
Solve the equation (a) Graphically, (b) Numerically, (c) Symbolically. Then solve the related inequality. |4x4 = 8, 4x 4 8
-
Normal profit is best described as: A. zero economic profit. B. total revenue minus all explicit costs. C. the sum of accounting profit plus economic profit.
-
Which of the following statements best explains why indifference curves are generally convex as viewed from the origin? A. The assumption of nonsatiation results in convex indifference curves. B. The...
-
The component least likely to be included in a measurement of gross domestic product (GDP) is: A. the value of owner-occupied rent. B. the annual salary of a local police officer. C. environmental...
-
A child indicates that she prefers going to the zoo over the park and prefers going to the beach over the zoo. When given the choice between the park and the beach, she chooses the park. Which of the...
-
In an industry comprised of three companies that are small-scale manufacturers of an easily replicable product unprotected by brand recognition or patents, the most representative model of company...
-
The market demand curve for heroin is said to be highly inelastic. Heroin supply is also said to be monopolized by the Mafia, which we assume to be interested in maximizing profits. Are these two...
-
In Exercises 105108, evaluate each expression without using a calculator. log(ln e)
-
Write structural formulas for the major organic products from each of the following reactions. (a) (b) (c) (d) (e) (1) KCN (2) H20, H,SO, (cat.) CN (1) DIBAL-H (1) CH,MgBr (2) H,O NH H,0, H.SO, (cat)...
-
Provide a detailed mechanism for each of the following reactions. (a) (b) (c) H,so, (cat.) OCH3 + CH OH (excess) 0 EtNH2 (excess) Cl NH H,O, H,SO, (cat.) OH
-
On heating, cis-4-hydroxycyclohexanecarboxylic acid forms a lactone but trans-4-hydroxycyclohexanecarboxylic acid does not. Explain.
-
An all-equity firm is considering the purchase of a depreciable asset that costs $680,000 and would be fully depreciated over six years using the straight-line method. The asset is expected to...
-
A firm owns three department stores. Each store has a debt-equity ratio of 16 percent and makes interest payments of $44,000 at the end of each year. The cost of the firm's levered equity is 18...
-
Belgian Bite Corp. is considering a project that has the following cash flow data. What is the project's IRR? Year 0 1 2 3 Cash flows $1,100 $450 $470 $490

Study smarter with the SolutionInn App


