Clomipramine is marketed under the trade name Anafranil and is used in the treatment of obsessive compulsive
Question:
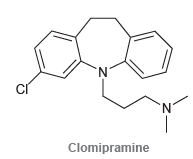
(a) Identify which nitrogen atom in clomipramine is more basic, and justify your choice.
(b) Draw the form of clomipramine that is expected to predominate at physiological pH.
Transcribed Image Text:
N. N' Clomipramine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
a The lone pair that is farthest away ...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A particularly strong and rigid polyester used for electronic parts is marketed under the trade name Glyptal. It is a polymer of terephthalic acid and glycerol. Draw a segment of the polymer, and...
-
Benzphetamine is an appetite suppressant that is marketed under the trade name Didrex and used in the treatment of obesity. Identify at least two different ways to make benzphetamine via a reductive...
-
Amobarbital is a sedative marketed under the trade name Amytal®. Propose a synthesis of amobarbital, using diethyl malonate and urea as two of the starting materials. CH3 CH2CH CHCH3 HN CH2CH3...
-
You have recently been hired as a fund manager in the portfolio management team of a bank. Your Director, John Tan has arranged a meeting next week to present an investment proposal to a prospective...
-
1. In a recent survey, people were asked whether they would prefer to work flexible hours-even when it meant slower career advancement-so they could spend more time with their families. The figure...
-
What do you think of the term medical error as a descriptor of adverse medical outcomes? After all, there are many medical procedures (e.g., invasive surgeries) and treatments (e.g., chemotherapy)...
-
The actual confidence level conf_level does not equal the theoretical confidence level 0.01 in Example 3.2. Explain why. Data from Example 3.2 Data form Figure 3.3 Example 3.2 (Confidence interval...
-
1. Under what conditions would it be practical for a company to send recruiters to college campuses to interview prospective employees, and when would it be impractical? What kinds of companies would...
-
The position of a particle moving along the x-axis is given by x(t) = = 4.2 2.5t m. (Assume t is in seconds.) (a) At what time (in s) does the particle cross the origin? 1.68 S (b) What is the...
-
The amount of time that a watch will run without having to be reset is a random variable having an exponential distribution with = 120 days. Find the probabilities that such a watch will (a) Have to...
-
Spermine is a naturally occurring compound that contributes to the characteristic odor of semen. Classify each nitrogen atom in spermine as primary, secondary, or tertiary. NH2 H,N 'N' Spermine
-
Draw the structures of all isomeric amines with molecular formula C 6 H 15 N that are not expected to produce any signal above 3000 cm 1 in their IR spectra.
-
Using Fermat's theorem, find \(3^{201} \bmod 11\).
-
Vedanta Resources has invested 8 million in a new mining project lasting 3 years. Depreciation is charged on a straight line basis to zero over the course of the project. The project generates...
-
The multinational media company News Corporation has a dual class share structure. Why might the company do this? Are there any drawbacks?
-
Your firm is considering purchasing a machine which requires an annual investment of 16,000. Depreciation is calculated using 20 per cent reducing balance (i.e. instead of depreciating the machine by...
-
If a firm is to cut costs as a result of falling revenues, how would this appear in the statement of financial position? Explain.
-
Explain the difference between dealer and agency markets. Why do you think both types of markets exist? Is there one type of market that is the best? Explain.
-
Convert the following hex numbers to binary: 15FD 16 ; 26EA 16 .
-
Suppose a population of bacteria doubles every hour, but that 1.0 x 106 individuals are removed before reproduction to be converted into valuable biological by-products. Suppose the population begins...
-
Draw the most stable conformation of: (a) ethylcyclohexane (b) 3-isopropyl-1,1-dimethylcyclohexane (c) cis-1-tert-butyl-4-isopropylcyclohexane
-
(a) Draw both chair conformations of cis-1,4-dimethylcyclohexane, and determine which conformer is more stable. (b) Repeat for the trans isomer. (c) Predict which isomer (cis or trans) is more stable.
-
Use your results from Problem 3-25 to complete the following table. Each entry shows the positions of two groups arranged as shown. For example, two groups that are trans on adjacent carbons...
-
Develop a simple application of two- variable linear programming problem to find an optimal product mix in a 'realistic' context. Specify the resources to be considered requirements, and the market...
-
According to the Google, Inc. Privacy Policy Litigation, on what date did Google announce their new universal privacy policy?
-
alcolm and Brianna were married on December 15, 2022. They spent the remainder of the tax year in Paris, France. Malcolm is a lifelong resident of North Carolina. Brianna was a lifelong resident of...

Study smarter with the SolutionInn App


