Compare the following three isomeric dienes: (a) Which compound will liberate the least heat upon hydrogenation with
Question:
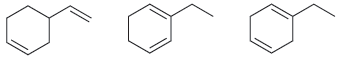
(a) Which compound will liberate the least heat upon hydrogenation with 2 mol of hydrogen gas? Why?
(b) Which compound will liberate the most heat upon hydrogenation with 2 mol of hydrogen gas? Why?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
a The conjugated diene will liberate the least heat because it is the most stabl...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
51+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What spectroscopic method could you use to distinguish among the following three isomeric acids? Tell what characteristic features you would expect for eachacid. CH3(CH213CO2H (CH/2CHCH-cO2H...
-
Solid NaI is slowly added to a solution that is 0.010 M in Cu1 and 0.010 M in Ag1. (a) Which compound will begin to precipitate first? (b) Calculate [Ag1] when CuI just begins to precipitate. (c)...
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
= Adobe Reader Touch Type here to search Active Research: Toyota's Hybrid Offer One of the most successful hybrid cars (cars that run on both battery and gasoline) is the Prius by Toyota. Visit...
-
What are three examples of data models? Which one is the most popular type?
-
For the case in exercise 6, write down the explicit equations for the wave function outside the geometrical optics approximation, and find a way to introduce the next-order corrections to the WKB...
-
In the field of ecology, the relationship between the concentration of available dissolved organic substrate and the rate of uptake (velocity) of that substrate by heterotrophic microbial communities...
-
The following transactions of Harmony Music Company occurred during 2010 and 2011: 2010 Mar 3 Purchased a piano (inventory) for $70,000, signing a six-month, 4% note payable. May 31 Borrowed $75,000...
-
As a network engineer for Kamehameha Institute, you will need to become familiar with the Packet Tracer simulation tool from Cisco. Packet Tracer is used to simulate and test network configurations....
-
Make a new version of your C++ program that creates instead a 2D array of 5x5 values of type boolean. Suppose indices represent people and that the value at row i , column j of a 2D array is true...
-
Identify each of the following quotes as being an example of either: the coordination problem, the invisible hand, creative destruction, or the incentive problem. a. "If you compare a list of todays...
-
True or False: Households sell finished products to businesses.
-
1. Lone Star Company would account for this as: A. A capital lease. B. A direct financing lease. C. A sales type lease. D. An operating lease. 2. What is the net carrying value of the lease liability...
-
On December 31, 2024, when its Allowance for Doubtful Accounts had a debit balance of $1,371, Sandhill Co. estimates that 8% of its accounts receivable balance of $77,800 will become uncollectible...
-
In January 2 0 2 2 , Khors Company issued nonqualified stock options to its CEO, Jenny. Because the company did not expect Jenny to leave the company, the options vested at the time they were granted...
-
Dozier Company produced and sold 1 , 0 0 0 units during its first month of operations. It reported the following costs and expenses for the month: Direct materials $ 8 9 , 0 0 0 Direct labor $ 4 5 ,...
-
Waveney DIY Centers ( WDC ) operates a few dozen stores in the eastern United States. The stores are popular with home remodelers, contractors, and do - it - yourself customers. The managers at...
-
Justin Timberlake, a single taxpayer, earns $ 7 6 , 6 0 0 in taxable income and $ 1 1 , 7 0 0 in interest from an investment in City of Los Angeles bonds. ( Use the U . S . tax rate schedule. )...
-
Use the following information concerning the age distribution of U.S. residents, based on 2012 population data. The data are rounded to the nearest million people. If one of these individuals is...
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
What product would you expect to obtain from addition of Cl2 to 1, 2-dimcthyl- cyclohexane? Show the stereochemistry of the product.
-
Addition of HC1 to 1, 2-dimethylcyclohexene yields a mixture of two products. Show the stereochemistry of each, and explain why a mixture is formed.
-
What product would you expect from the reaction of cyclopentane with NBS and water? Show the stereo chemistry.
-
Apply Euler's method by hand with N = 4 to find yo, Y1, Y2, Y3, Y4 on [0,1] where Y' y' = - 2y + 3t y(0) =1 Note: Please don't use Python for this. You are allowed to use a calculator do the...
-
What amount paid on September 6 is equivalent to $ 2 , 6 0 0 paid on the following December 1 if money can earn 6 . 6 % ? ( Use 3 6 5 days a year. Do not round intermediate calculations and round...
-
For R1= 6.8k, and R2= 1k, Then: V(1) = v V. 1(2) = mA.

Study smarter with the SolutionInn App


