Consider the following reaction: a. Draw the mechanism of this reaction. b. What is the rate equation
Question:
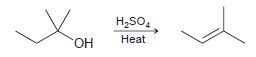
a. Draw the mechanism of this reaction.
b. What is the rate equation of this reaction?
c. Draw an energy diagram of the reaction.
Transcribed Image Text:
H2SO, Heat OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a b This is an E1 pro...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Consider the following reaction at equilibrium: From the data shown here, calculate the equilibrium constant (both KP and Kc) at each temperature. Is the reaction endothermic or exothermic?...
-
Consider the following reaction at 25C: Fe(OH)2 Fe2+(aq) + 2OH-(aq) Calculate G for the reaction. Ksp for Fe(OH)2 is 1.6 10-14.
-
rewrite/downside Integrity and credibility are the ethics of professional practice that Juan Gomez was lacking in this instance. Juan Gomez lacked integrity because he created a conflict of interest...
-
Ethical Considerations" Please respond to the following: Reflect upon the responsibilities placed on auditors by the PCAOB, and discuss whether those expectations are adequate considering current...
-
The data in the following table represent the proportion of Americans 25 years of age or older at various levels of educational attainment in 2013. If we let M represent the event that a randomly...
-
What we do if there are too many steps in a normal flow?
-
Set up T accounts for Cash, Unearned Ticket Revenue, and Ticket Revenue. Post the following two transactions to the appropriate accounts, indicating each transaction by letter: (a) Sold 1,200 season...
-
A set S R^n is convex if for all x, y S and t [0, 1], we have (1 t)x + t y S (in other words, if S contains the points x and y, it contains the entire line segment between them). (a) If W R^n is a...
-
A 450-mm-long AISI 1020 steel rod is subjected to a tensile load of 55 kN. The allowable tensile stress is 140 MPa and the allowable total elongation is not to exceed 0.2 mm. Calculate the required...
-
A purely competitive firm whose goal is to maximize profit will choose to produce the amount of output at which: a. TR and TC are equal. b. TR exceeds TC by as much as possible. c. TC exceeds TR by...
-
If it is possible for a perfectly competitive firm to do better financially by producing rather than shutting down, then it should produce the amount of output at which: a. MR < MC. b. MR = MC. c. MR...
-
What are ADRs?
-
Public financial management ( PFM ) has been narrowly defined to cover the budget cycle, which typically centers around ( 1 ) budget formulation; ( 2 ) budget execution; ( 3 ) accounting and...
-
On 1 5 October 2 0 1 9 , the board of directors of MAA Bhd decided to close down one of its overseas divisions due to continuous losses. For this reason, a detailed plan was drafted and all costs...
-
Lusanda. When the parties married, Sipho had no assets apart from his interest in his pension fund, valued at R50 000. He had started a new business and had an outstanding loan of R100 000. Lusanda...
-
Metha was employed by Safaricom _Plc at a basic salary of ksh 18 million per annum. The service level agreements sated that Metha was eligible for (i) house allowance of ksh 200,000 per month (ii)...
-
Current Attempt in Progress Flint Company sold equipment on July 1, 2021 for $78,500. The equipment had cost $235,000 and had $139,000 of accumulated depreciation as of January 1, 2021. Depreciation...
-
Evaluate the expression. (a) 8 C 0 (b) 8 P 0
-
Create an appropriate display of the navel data collected in Exercise 25 of Section 3.1. Discuss any special properties of this distribution. Exercise 25 The navel ratio is defined to be a persons...
-
In the DebyeHckel theory, the counter charge in a spherical shell of radius r and thickness dr around the central ion of charge +Q is given by Q 2 re r dr. Calculate the radius at which the counter...
-
Calculate the solubility of CaCO 3 (K sp = 3.4 10 -9 ) a. In pure H 2 O. b. In an aqueous solution with I = 0.0250 mol kg 1 . For part (a), do an iterative calculation of and the solubility until...
-
Calculate the probability of finding an ion at a distance greater than 1/ from the central ion.
-
Aaron, Deanne, and Keon formed the Blue Bell General Partnership at the beginning of the current year. Aaron and Deanne each contributed $138,000, and Keon transferred an acre of undeveloped land to...
-
The following information pertains to the inventory of Parvin Company: Jan. 1 Apr. 1 Oct. 1 Beginning inventory Purchased Purchased 400 units @ 2,400 units @ 1,100 units $17 $22 $23 During the year,...
-
Gold Nest Company of Guandong, China, makes birdcages for the South China market. The company sells its birdcages through an extensive network of street vendors who receive commissions on their...

Study smarter with the SolutionInn App


