Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1
Question:
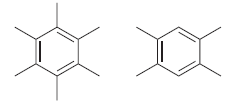
a) IR spectroscopy?
b) 1H NMR spectroscopy?
c) 13C NMR spectroscopy?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
a The first compound would lack CH stretching signals just above 3000 cm 1 while the seco...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you distinguish between the following compounds using 13 C NMR spectroscopy?
-
How would you distinguish between each pair of compounds using mass spectrometry? a. b. N.
-
How would you distinguish between each pair of compounds using high-resolution mass spectrometry? a. b. .
-
Prepare journal entries to record each of the following sales transactions of TFC Merchandising. TFC uses a perpetual inventory system and the gross method. May 1 9 Sold merchandise for $600, with...
-
What advantages and disadvantages does offering trade credit provide to a small business?
-
If you have $50,000 in an interest-bearing savings account that pays 2 percent annual interest, how much interest will you earn during a 30 day month?
-
You rolled a six-sided die 60 times and got the following tally. 20 ones 20 twos 15 threes 3 fours 2 fives 0 sixes Does this seem like a reasonable result? What inference might you draw from the...
-
Reconsider the Leisure Airlines problem from Section 12.1. The demand forecasts shown in Table represent Leisure Airs best estimates of demand. But because demand cannot be forecasted perfectly, the...
-
a. Draw a flowchart of a program called CalculateAverage that asks the user to enter 3 integers (using a loop), then calculates and displays the average of the numbers. b. Trace the execution of the...
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
Consider the equilibrium C 2 H 6 (g) C 2 H 4 (g) + H 2 (g). At 1000.K and a constant total pressure of 1.00 bar, C 2 H 6 (g) is introduced into a reaction vessel. The total pressure is held constant...
-
The following compound cannot be made with either a Friedel-Crafts alkylation or acylation. Explain.
-
Kids 'n Caboodle, a children's clothing store, had the following cash receipts and disbursements for its first year of operations: The store has no accounts receivable (it accepts only cash or bank...
-
Think of recent events from the sports industry that show a taste for discrimination. Do they represent consumer, employer, or employee discrimination?
-
You have been asked ro design a series of user interface screens that will be used by both employees and customers. You proudly roll out your prototypes, expecting accolades and high pratse. Instead,...
-
A company expects FCF of 2$10 million at Year 1 and FCF of $20 million at Year 2; after Year 2, FCF is expected to grow at a 5% rate. If the WACC is 10%, then what is the horizon value of operations,...
-
Use what you know about the prisoners dilemma to explain why the English Premier League teams such as Arsenal have so many foreign players even when, as a group, the Premier League teams agree that...
-
A company expects to have FCF of $600 at Year 10, which is expected to grow at a constant rate of 4% thereafter. If the WACC is 8%, what is the value of operations at Year 10, HV10? ($15,600)
-
Use synthetic division to perform each division. x5 + 1 +1
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
Give the products of the reaction of 2-ethyl- 2-methyloxirane (or other compound indicated) with each of the following reagents. (a) Water, H3O+ (b) Water, NaOH, heat (c) NA+ CH3O- in CH3OH (d) CH3OH...
-
The Clifford Corporation has announced a rights offer to raise $36 million. The stock currently sells for $28 per share and there are 18 million shares outstanding. If the subscription price is set...
-
Superman Enterprises has just completed an initial public offering. The firm sold 4,700,000 new shares at an offer price of $17.00 per share. The underwritering spread was $0.78 a share. The firm...
-
ABV is a non-dividend paying stock whose current price is $50. Its volatility is 12%. Over each of the next two 6-month periods the stock price is expected to go up by 9% or down by 8%. The risk-free...

Study smarter with the SolutionInn App


