Consider the structures of 2-nitrophenol and 3-nitrophenol. These compounds have very different pKa values. Predict which one
Question:
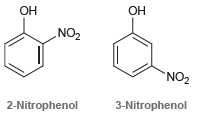
Transcribed Image Text:
ОН ОН „NO2 `NO2 2-Nitrophenol 3-Nitrophenol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
2nitrophenol is expected to be more acidic lower pK a ...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds, predict which one has the higher molecular dipole moment, and explain your reasoning. (a) Ethyl chloride or ethyl iodide (b) 1-bromopropane or cyclopropane (c) Cis-2,...
-
A bicycle chain consists of a series of small links, each 12 mm long between the centers of the pins (see figure). You might wish to examine a bicycle chain and observe its construction. Note...
-
Consider the following four structures: a. Which of these compounds have the same physical properties (melting point, boiling point, density, and so on)? b. Which of these compounds are trans...
-
How do the Uniform Trade Secrets Act (UTSA) and the Economic Espionage Act of 1996 differ? Why don't these acts always provide a sufficient remedy for the theft of trade secrets?
-
Add support for a ListIterator to the MyLinkedList class, as was done in Exercise 3.13. Add support for a ListIterator to the MyArrayList class. The ListIterator interface in java.util has more...
-
The table below lists measured amounts of redshift and the distances (billions of light-years) to randomly selected clusters of galaxies. For the prediction interval, use a 90% confidence level with...
-
A reactor is to be designed for the oxidation of sulfur dioxide, with excess oxygen from air, to sulfur trioxide. The entering feed, at \(550 \mathrm{~K}\) and \(1.1 \mathrm{bar}\), consists of...
-
A new CEO was hired to revive the floundering Champion Chemical Corporation. The company had endured operating losses for several years, but confidence was emerging that better times were ahead. The...
-
The loading Code AS1170.1 requires that the reading room (without a bookstore) at the library be designed for a floor live load of Q = 2.5 kPa. If the spans between two steel beams are L = 12 m as...
-
The individual financial statements for Gibson Company and Keller Company for the year ending December 31, 2011, follow. Gibson acquired a 60 percent interest in Keller on January 1, 2010, in...
-
Consider the following substitution reaction: a) Determine whether this reaction proceeds via an S N 1 or S N 2 process. b) Draw the mechanism of this reaction. c) What is the rate equation of this...
-
Draw a Lewis structure for each of the compounds below: a) CH 2 = CHOCH 2 CH(CH 3 ) 2 b) (CH 3 CH 2 ) 2 CHCH 2 CH 2 OH c) (CH 3 CH 2 ) 3 COH d) (CH 3 ) 2 C = CHCH 2 CH 3 e) CH 2 = CHCH 2 OCH 2 CH(CH...
-
Software games company, Avalanche Entertainment, is considering expanding its highly successful online game franchise to the board game or trading card environment. The company has decided that it...
-
Lisa Tylor, CFO of Purple Rain Co., concluded from the Baumol model that the optimal cash balance for the firm is $10 million. The annual interest rate on marketable securities is 5.8 per cent. The...
-
While charging a general admission price of HK$10 for adults most days of the week, the Hong Kong Museum of History offers free admission every Wednesday. Why do you suppose that museums often price...
-
In 2016, Italy, which is a highly indebted nation, sold 50-year bonds at a yield of 2.8 percent, and buyers were given access to 5 billion euros in the form of these bonds. Would you consider this a...
-
You have just won a million-dollar lottery and will receive $50,000 at the end of each year for 20 years. What is the present value of the 20 annual payments to you today?
-
Explain what will happen to the present value of money one year from now if the market interest rate falls? What if the market interest rate rises?
-
Identify the managers by level and area at your college or university.
-
The tractor is used to lift the 150-kg load B with the 24-mlong rope, boom, and pulley system. If the tractor travels to the right at a constant speed of 4 m/s, determine the tension in the rope when...
-
Rank the compounds in each group according to their reactivity toward electrophilic substitution. (a) Chlorobenzene, o-dichlorobenzene, benzene (b) p-Bromonitrobenzene, nitrobenzene, phenol (c)...
-
A Predict the major mono-alkylation products you would expect to obtain from reaction of the following substances with chloromethane arid AlCl3: (a) Bromobenzene (b) m-Bromo-phenol (c)...
-
Name and draw the major product(s) of electrophilic chlorination of the following compounds: (a) in-Nitro phenol (b) o-Xylene (c) p-Nitro benzoic acid (d) p-Bromo benzenesulfonic acid
-
A 9 Loan amount Annual interest rate Number of years until maturity Annual payment amount B $300,000.00 Date Graded Worksheet 7% 4 On January 1, 2024, a company borrows cash from a local bank by...
-
As we have seen in this course, virtually every generation has felt that the existing threat to its privacy was unprecedented in nature. That said, the sheer capacity to collect metadata which exists...
-
In 1982 the inflation rate hit 16%. Suppose that the average cost of a textbook in 1982 was $20. What was the expected cost in the year 2017 if we project this rate of inflation on the cost? (Assume...

Study smarter with the SolutionInn App


