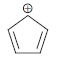
Draw a Frost circle for the following cation, and explain the source of instability of this cation.
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Six electrons are r...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use a Frost circle to determine the -electron structure of the cyclopropenyl cation, which has two electrons cyclopropenyl cation
-
The cyclopropenyl cation has a three-membered ring that contains a continuous system of overlapping p orbitals. This system contains a total of two Ï electrons. Using a Frost circle, draw an...
-
(a) The following diagram represents the reaction of PCl4+ with Cl-. Draw the lewis structures for the reactants and products, and identify the lewis acid and the lweis base in the reaction. (b) The...
-
Solve each system using the substitution method. If a system is inconsistent or has dependent equations, say so. -5x + 2y = -2 x + 6y = 26
-
The final project for this course is a case study in which you will select a real-world accounting ethics case involving earnings mismanagement, fraud, poor internal controls, or poor corporate...
-
You decide to open an individual retirement account (IRA) at your local bank that pays 9 percent/year/month. For the next 30 years, starting 1 month from today, you will deposit $200 per month into...
-
Refer to Exercise 4.2. (a) Determine the cumulative probability distribution \(F(x)\). (b) Graph the probability distribution of \(f(x)\) as a bar chart and below it graph \(F(x)\). Data From...
-
Data on sales revenue Y, television advertising expenditures X1, and print media advertising expenditures X2 for a large retailer for the period 1988-1993 were given in Problem 11 of Chapter 8. Use...
-
Required information [The following information applies to the questions displayed below.] George and Wanda received $30,500 of Social Security benefits this year ($11,700 for George; $18,800 for...
-
Consider the extended LAN connected using bridges B1 and B2 in Fig. 4-41(b). Suppose the hash tables in the two bridges are empty. List all ports on which a packet will be forwarded for the following...
-
How many signals do you expect in the 13 C NMR spectrum of each of the following compounds? a. b. c. d. Br
-
Do you expect the following dianion to exhibit aromatic stabilization? Explain.
-
Explain fully what you believe are the most important features in the successful implementation and management of organisational change.
-
USAA is a Fortune 500 insurance and financial services company with 2016 annual revenues exceeding $27 billion. The company was founded in 1922 by 25 Army officers who decided to insure each others...
-
What are the order winners and order qualifiers for WalMart? Toyota? BMW? Sony?
-
Take a look at the following for-loop: Without running this code on the command window, write out what the print statement would be at each iteration. int index = 6; for (int i = index + 1; i < index...
-
Deming suggests that 94% of quality problems are a function of the design of the system (the common causes of error) and therefore the responsibility of management to resolve. What barriers to their...
-
Describe briefly the format of the standard audit report currently in use in the United Kingdom (as required by ISA (UK and Ireland) 700 (FRC, 2013b) and how it differs from the standard audit report...
-
What are fission fragments, and why are they so dangerous?
-
Synthesize the products by drawing out reagents and intermediates along the way. `N H. OH HO HO
-
Give the products (if any) expected when the compounds in Problem 5.13 are treated with ozone followed by aqueous hydrogen peroxide.
-
In each case, give the structure of an eight-carbon alkene that would yield each of the following compounds (and no others) after treatment with ozone followed by dimethyl sulfide. (b)...
-
What aspect of alkene structure cannot be determined by ozonolysis?
-
This project requires an investment in fixed assets, at time 0, of 44,000,000. Assets will be depreciated straight line in 5 years. This project has a 4 year life. The revenues for each of the 4...
-
Lorenzo Company applies overhead to jobs on the basis of direct materials cost. At year - end, the Work in Process Inventory account shows the following. Work in Process Inventory Date Explanation...
-
T-bill with a $10,000 par has 174 days until maturity. The T-bill has a bank discount bid quote of 2.208% and ask quote of 2.198%. What is the return on the investment in this T-bill if the trader...

Study smarter with the SolutionInn App


