Draw a resonance structure for each of the compounds below. a. b. c. N
Question:
a.
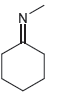
b.
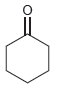
c.
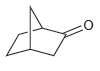
Transcribed Image Text:
N°
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
a ...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a second resonance structure for each of the following carbocations: (a) (b) (c) CH3CH CHH2 ( 3)2 4
-
Draw a resonance structure of the acetonitrile anion, :CH 2 C N, and account for the acidity of nitriles.
-
Draw a resonance structure of the compound below, which was isolated from the fruits of Ocotea corymbosa, a native plant of the Brazilian Cerrado.
-
The net income for Rips Curling Corp. for the year ended December 31, 2021, was $700,000. Rip had 50,000 ordinary shares outstanding at the beginning of the year. Rip declared and distributed a...
-
When translated into a project network, a work package will become A single activity A critical path Over or more activities An arrow A milestone?
-
Lower bound: 21 , upper bound: 29 Determine the point estimate of the population mean and margin of error for each confidence interval.
-
The probability that a wafer contains a large particle of contamination is 0.01. If it is assumed that the wafers are independent, what is the probability that exactly 125 wafers need to be analyzed...
-
A bank has written European a call option on one stock and a European put option on another stock. For the first option, the stock price is 50, the strike price is 51, the volatility is 28% per...
-
Caleb, a high school wood and metal work teacher, has provided details of the following transactions during the year ended 3 0 June 2 0 2 3 : - Recieved salary of $ 1 0 4 , 0 0 0 for the school he is...
-
A company sells precision grinding machines to four customers in four different countries. It has just signed a contract to sell, two months from now, a batch of these machines to each customer. The...
-
A cup of water at 278 K (the system) is placed in a microwave oven and the oven is turned on for 1 minute during which it begins to boil. State whether each of q, w, and U is positive, negative, or...
-
In the experiment shown in Figure 2.4a and 2.4b, ÎU surroundings < 0, but ÎT surroundings > 0. Explain how this is possible. Figure 2.4 Electrical generator Mass Heating- coil Bunsen...
-
In the same 2012 survey conducted in Exercise 43, Gallup reported that 66% of American investors say that the federal budget deficit is hurting the U.S. investment climate a lot. Is there any...
-
3. Consider two countries with the following characteristics. Country A has no restrictions on bank branching and banks in Country A are permitted to offer investment and insurance products along...
-
The Swifty Hat Shop Limited counted the entire inventory in its store on August 3 1 and arrived at a total inventory cost of $ 9 3 , 9 0 0 . The count included $ 6 , 9 0 0 of inventory held on...
-
Ace Inc., a semiweekly depositor, pays wages on Thursday, September 3 0 , that gives rise to a deposit obligation of $ 7 2 , 0 0 0 . On Friday, October 1 , Ace Inc. pays additional wages that give...
-
On January 1 , 2 0 2 0 , Kendall Co . purchased to hold to maturity, 1 0 0 0 , $ 1 , 0 0 0 , 9 % bonds for a price to yield an effective interest rate of 1 0 % . Interest is paid semiannually on...
-
Lancaster Company had earnings net of tax but before extraordinary items of $ 5 0 0 , 0 0 0 . They had an extraordinary loss net of tax of $ 1 0 0 , 0 0 0 . Lancaster had a weighted average number of...
-
Describe how the Bohr model may be used to account for characteristic x-ray spectra in heavy atoms.
-
Consider model (9.18). What is the effect on the model parameter estimates, their standard errors, and the goodness-of-fit statistics when (a) The times at risk are doubled, but the numbers of deaths...
-
When, in separate reactions, 2-methylpropene, propene, and ethene are allowed to react with HI under the same conditions (i.e., identical concentration and temperature), 2-methylpropene is found to...
-
Propose a mechanism that accounts for the following reaction. HI
-
When 3, 3-dimethyl-2-butanol is treated with concentrated HI, a rearrangement takes place. Which alkyl iodide would you expect from the reaction? (Show the mechanism by which it is formed.)
-
Describe, in steps, at least one strategy used to attack embedded operating systems. Describe, in steps, at least one strategy used to defend against the chosen attack.
-
how does a hypercompetetice environment challenge those in an industry
-
Describe how managers can reduce unnecessary costs, including real - world examples of how it has been done

Study smarter with the SolutionInn App


