Firefly luciferin is the compound that enables fireflies to glow. a. The structure exhibits three rings. Identify
Question:
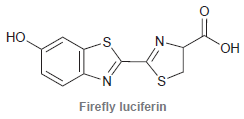
a. The structure exhibits three rings. Identify which of the rings are aromatic.
b. Identify which lone pairs are participating in resonance.
Transcribed Image Text:
Но ОН N. Firefly luciferin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a b One of the lone ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Exposing Photographic Film the light-sensitive compound on most photographic films is silver bromide, Ag Br. A film is "exposed" when the light energy absorbed dissociates this molecule into its...
-
Experiments during a recent summer on a number of fireflies (small beetles, Lampyridae photinus) showed that the average interval between flashes of individual insects was 16.3 s at 21.0oC and 13.0 s...
-
Compound A with molecular formula C6H10 has two peaks in its spectrum, both of which are singlets (with ratio). Compound A reacts with an acidic aqueous solution containing mercuric sulfate to form...
-
Correction, overproduction, inventory and motion are all examples of: a. Waste b. 5 S target areas c. Noise d. Value-added activities
-
What mistakes are first-time exporters most likely to make? Outline the steps a small company should take to establish a successful export program.
-
The Allison-Chambers Corporation, manufacturer of tractors and other heavy farm equipment, is organized along decentralized product lines, with each manufacturing division operating as a separate...
-
What is the most accepted input power required for an ICP accelerometer?
-
Farley Company identifies the following items for possible inclusion in the taking of a physical inventory. Indicate whether each item should be included or excluded from the inventory taking. (a)...
-
4. Evaluate the following limits, if they exist. If a limit does not exist, state DNE. For these limits, recall that lim tan x a. lim X-0 x sin x COS X-1 = 1 and lim = 0. X-0 x X40X b. lim sin(x-1)...
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
Identify which of the following compounds are aromatic. a. b. c. d. e. O.
-
Identify each of the following compounds as aromatic, nonaromatic, or antiaromatic. Explain your choice in each case. a. b. c. d. e. f. g. h. :N-H
-
Find the normal approximation for the binomial probability P(x > 9), where n = 13 and p = 0.7. Compare this to the value of P(x > 9) obtained from Table 2.
-
List the benefits an Israeli firm can have by listing its stock on the NASDAQ stock exchange in the United States, in addition to a listing on the local Israeli exchange. Can you see any drawbacks to...
-
The safe minimum standard (SMS) is a special case of the mainstream economics conception of sustainability. Can you present an argument that attempts to validate this statement?
-
Most companies use the indirect method of preparing a cash flow statement. You are invited to meet a finance director of a leading listed company which uses the indirect method. Suggest two arguments...
-
In what specific ways would trade among nations complicate the effort to construct an environmentally adjusted sustainable national income accounting? Explain.
-
You are the fund manager in charge of the investment portfolios of wealthy private clients. One of them has asked you to explain the statement of cash flows that appears in the annual report of a...
-
Solve each problem The circumference of a circle varies directly as the radius. A circle with radius 7 in. has circumference 43.96 in. Find the circumference of the circle if the radius changes to 11...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
Complete the following reactions. If no reaction is tikety, explain why. (a) (b) CH,SH + NaOH -_, (1 equiv.) 25 C CH OH
-
Outline a synthesis of each ether using either alcohol dehydration or alkene addition, as appropriate. (a) 2-methoxy-2-methylbutane (b) dibutyl ether
-
Give the structure of the that would with mCPBA to give each of the following expoxides. (a) (b) . /A C CH2 H,C C-4 CH,
-
Could Arthur Murray have created language that would have avoided the application of impossibility of performance? What if Parker was not incapac-itated but that it was simply painful to dance after...
-
In the text's case on the county clerk and marriage licenses, whatare the ethical issues involved? What are the conflicting ethical values and whichvalues were maximized/minimized? The Case of the...
-
2 Altira Corporation provides the following information related to its inventory during the month of August 2024: August 1 Inventory on hand-2,000 units%;B cost $5.30 each. August 8 Purchased 8,000...

Study smarter with the SolutionInn App


