For each of the following alkenes, assign the configuration of the double bond as either E or
Question:
a.
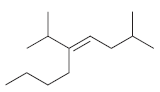
b.
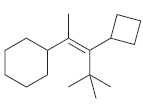
c.
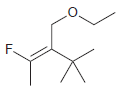
d.
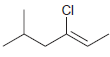
Transcribed Image Text:
F.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (17 reviews)
a...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The second-order rate constant (in units of M-1 s-1) for acid-catalyzed hydration at 25°C is given for each of the following alkenes: a. Calculate the relative rates of hydration of the alkenes....
-
Name each of the following alkenes or alkynes. a. CH2 = CH-CH2-CH3 b. c. d. e. f. g. CH3 C-CH-CH3 CH3 CH CH3 CH3CH2CH CH CH CH CH3 CH, C-CH-CH CH, CH2-CH, CH3 CH2CHs CH, CH2CH3 CH3 C C-CH CH3 CH3
-
Explain why each of the following alkenes is stable or unstable. (a) 1,2-dimethylcyclopentene (b) trans-1,2-dimethylcyclopentene (c) trans-3,4-dimethylcyclopentene (d) trans-1,2-dimethylcyclodecene...
-
In your opinion, was Saks' zero tolerance policy for employee theft reasonable? Was the policy likely cost-effective? Defend your answers.
-
What happens when a person has too much money? What about when an economy has too much money?
-
Consider the state transition diagram of Figure. Suppose that it is time for the OS to dispatch a process and that there are processes in both the Ready state and the Ready/Suspend state, and that at...
-
Define the following : (a) Clearance volume (b) Swept volume (c) compression ratio
-
Answer the following multiple-choice questions. 1. GAAP stands for: (a) Governmental auditing and accounting practices. (b) Generally accepted attest principles. (c) Government audit and attest...
-
3 Consider the mass of the objects in the activity (small ball large ball, feather) and compare the gravitational force between them and the Earth. If distance is the same which object would you...
-
Arbitrators As a client of a brokerage firm, Howsam invested in four limited partnerships. These investments were made between 1986 and 1994. The client agreement signed by Howsam required all...
-
Which of the following best describes the efficiency of monopolistically competitive firms? a. Allocatively efficient by productively inefficient. b. Allocatively inefficient but productively...
-
Draw the mechanism and predict the product of the following reaction. In this case, H 3 O + must be used as a proton source instead of water. Explain why. 1) xs MeMgBr 2) H*
-
Explain the difference between a mean and a weighted mean.
-
Lake County Credit Union (LCCU) approved a mortgage loan to Giselle for $175,000 at a fixed rate of 3.75 percent with a thirty-year term secured by the home. After paying $10,500 of the mortgage,...
-
One who finds abandoned property acquires good title to the property against the whole world, except the true owner. (True/False)
-
Selma borrows $125,000 from Riverview Credit Union to buy a home. Among the terms that must be disclosed under federal law is the annual percentage rate. This rate is a. the actual cost of the loan...
-
An employer can dismiss an employee due to garnishment. (True/False)
-
If goods are confused due to a wrongful act and the innocent party cannot prove what percentage is theirs, all of the goods belong to the wrongdoer. (True/False)
-
Marta is 52 years of age and expects to retire from her current employer at age 65. Martas employer contributes $2,000 to a qualified retirement plan for Martas benefit in the current year. How will...
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
One of the two chair structures of cis-1-chloro-3-methylcyclohexane is more stable than the other by 15.5kJ/mol (3.7kcal/mol). Which is it? What is the energy cost of a 1, 3-diaxial interaction...
-
The German chemist J. Bredt proposed in 1935 that bicycloalkenes such as 1-norbornene, which have a double bond to the bridgehead carbon, are too strained to exist. Make a molecular model of...
-
Tell whether each of the following substituents on a steroid is axial or equatorial. (A substituent that is ?up? is on the top face of the molecule as drawn, and a substituent that is ?down? is on...
-
You anticipate the receipt of money in 200 days, which you will use to purchase stocks in a particular company. The stock is currently selling for $51 and will pay a $0.5 dividend in 50 days and...
-
1) Based on the stock chart for Michaels Companies Inc, what do you think the short and long-term growth potentials are for this company? (discuss the advantages/disadvantages) Link to the stock...
-
After being drafted in the first round of the NFL draft, a star defensive end invests his signing bonus of $9,827,000.00 in a mutual fund. The fund pays on average 7.00% APR. The player will not...

Study smarter with the SolutionInn App


