For each of the following carbocations determine if it will rearrange, and if so, draw the carbocation
Question:
a.
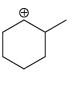
b.
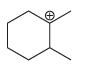
c.
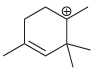
d.

e.

f.

g.
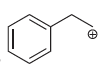
h.

Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a b This carbocation is tertiary and will not rearrange c d Th...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a second resonance structure for each of the following carbocations: (a) (b) (c) CH3CH CHH2 ( 3)2 4
-
Each of the following carbocations has the potential to rearrange to a more stable one. Write the structure of the rearranged carbocation. (a) CH3CH2CH2+ (b) (CH3)2CHC_HCH3 (c) (CH3)3CCHCH3 (d)...
-
Each of the following carbocations can rearrange to a more stable ion. Propose structures for the likely rearrangement products. H, (a) CH3CH2CH2CH2* (b) CH3CHCHCH3 CH CH CH2* (c)
-
Evaluate the geometric series or state that it diverges. 8 00 k=0 2k 75
-
The premium on a call option on the market index with an exercise price of 100 is $1.90 when originally purchased. After 2 months the position is closed and the index spot price is 102. If interest...
-
The weights of four randomly and independently selected bags of tomatoes labeled 5 pounds were found to be 5.1, 5.0, 5.3, and 5.1 pounds. Assume Normality. a. Find a 95% confidence interval for the...
-
An RVE from a particle-reinforced composite is shown in Figure 3.12. The particle has a cross-sectional area \(A_{\mathrm{p}}(x)\) that varies with the distance \(x\), and the stresses and strains in...
-
Phillips, Inc., a cash basis C corporation, completes $100,000 in sales for year 1, but only $75,000 of this amount is collected during year 1. The remaining $25,000 from these sales is collected...
-
On January 1, Sharp Company purchased $75,000 of Sox Company 6% bonds, at a time when the market rate was 5%. The bonds mature on December 31 in five years, and pay interest annually on December 31....
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Draw the curved arrows that accomplish each of the following transformations: a. b. c. H. H.
-
In each of the following cases compare the bonds identified with red arrows, and determine which bond you would expect to have the largest bond dissociation energy: a. b. CI .F Br
-
On the following page, you will find the condensed financial statements for High Inc. and Low Inc. for 2016. INSTRUCTIONS 1. Compute the following ratios for each company: a. Rate of return on net...
-
Explain the term customer service representatives (CSRs)?
-
In regard to time, how do the income statement and balance sheet differ?
-
Which of the following best represents a contractionary fiscal policy? A. Public spending on a high-speed railway B. A temporary suspension of payroll taxes C. A freeze in discretionary government...
-
List some characteristics of organizational buyers or buying centers.
-
a. Briefly describe the following health services settings: Hospitals Ambulatory care Home health care Long-term care Integrated delivery systems b. What are the benefits attributed to...
-
A profit-maximizing competitive firm that is making positive profits in long-run equilibrium (may/may not) have a technology with constant returns to scale.
-
Give codons for the following amino acids: (a) Th (b) Asp (c) Thr
-
When pseudoionone is treated with BF3 in acetic acid, ring closure takes place and α-and β-ionone are produced. This is the next step in the vitamin A synthesis. (a) Write...
-
(a) Write resonance structures for the anion of acetonitrile that account for its being much more acidic than ethane. (b) Give a step-by-step mechanism for the condensation of benzaldehyde with...
-
Starting with ketones and aldehydes of your choice, outline a directed aldol synthesis of each of the following using lithium enolates: (a) (b) (c) O OH CGH5 O OH
-
Fixed cost per unit is $7 when 25,000 units are produced and $5 when 35,000 units are produced. What is the total fixed cost when nothing is produced?
-
Menlo Company distributes a single product. The company's sales and expenses for last month follow: Total Per Unit Sales $316,000 $20 Variable expenses 221,200 14 Contribution margin 94,800 $ 6 Fixed...
-
Shown are selected financial data for Another World and Imports, Inc., at the end of the current year. Net credit sales Cost of goods sold Cash Another World Imports, Inc. $ 675,000 $560,000 504,000...

Study smarter with the SolutionInn App


