For each of the following compounds, determine whether or not you would expect its IR spectrum to
Question:
a.
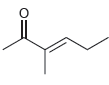
b.
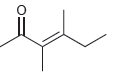
c.
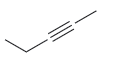 .
.
d.
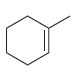
e.
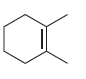
f.
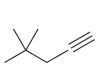
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a b No ...View the full answer

Answered By

Parvesh Kumar
I am an experienced Mathematics and Statistics tutor with 10 years of experience teaching students and working professionals. I love teaching students who are passionate to learn subjects or wants to understand any mathematics and statistics concept at graduation or master’s level. I have worked with thousands of students in my teaching career. I have helped students deal with difficult topics and subjects like Calculus, Algebra, Discrete Mathematics, Complex analysis, Graph theory, Hypothesis testing, Probability, Statistical Inference and more. After learning from me, students have found Mathematics and Statistics not dull but a fun subject. I can handle almost all curriculum of mathematics. I did B.Sc (mathematics), M.Sc (mathematics), M.Tech (IT) and am also Gate (CS) qualified. I have worked in various college and school and also provided online tutoring to American and Canadian students. I look forward to discussing with you and make learning a meaningful and purposeful
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds, give the systematic name and the common name (for those that have common names), and indicate whether the amines are primary, secondary, or tertiary: a. b. c....
-
For each of the following compounds, use the nitrogen rule to determine whether the molecular weight should be even or odd. Then calculate the expected m/z value for the molecular ion. a. b. c. d. O:...
-
1. Draw a flow chart for the following process You are approaching a local bank for getting an educational loan and draw a flow chart with all the process in detail and the decision node that you can...
-
A majority element in an array, A, of size N is an element that appears more than N/2 times (thus, there is at most one). For example, the array 3, 3, 4, 2, 4, 4, 2, 4, 4 has a majority element (4),...
-
The sample data below are changes in LDL cholesterol levels in clinical trials of Lipitor (atorvastatin). It was claimed that Lipitor had an effect on LDL cholesterol. Negative values represent...
-
An ideal gas, with \(C_{p}=7 \mathrm{cal} / \mathrm{mol}-{ }^{\circ} \mathrm{C}\), is compressed from 1 to \(50 \mathrm{~atm}\) while its temperature rises from 25 to \(150^{\circ} \mathrm{C}\). How...
-
The July 31, 2014, unadjusted trial balance for Daimler Tours after its first month of operations is shown below: Required 1. Enter the unadjusted trail balance onto a work sheet. 2. Using the...
-
Review the first 2 case studies: https://www.countyhealthrankings.org/ Synthesize each in a brief summary (use each of these as a subheading) of key concepts a. Characterizing a Community b....
-
A plant has water tank mounted on the top of a 27 m platform. The tank is 10 m high. Calculate the height of the water in the tank if a pressure gauge on the second floor at a height of 5 m from the...
-
An indicator of the degree of competition in an industry is the concentration ratio. It measures a. The percentage of sales in the industry by the largest firms. b. The percentage of profit in the...
-
Local telephone service was once an area in which consumers had no choices. Many young people no longer use landlines, preferring instead to use their cellular phones. This means that the market has...
-
For each of the following lists of premises, derive the indicated conclusion and complete the justification. For tautology, derive a conclusion that is simpler than the premise. 1. Ev E 2. AVA 3. G....
-
Briefly explain how the use of a cash register provides internal control over cash receipts.
-
From the following business transactions in July 2022 you are required to prepare comparative general journals for both perpetual and periodic inventory; post to the general ledger, where the...
-
At the end of the financial year, prepare the general journal to close out the sales account $32 000 and cost of sales account $19 500 to trading account and post to the general ledger.
-
From the following transactions of D Brad made in January 2022, you are required to prepare the general journals using perpetual inventory, post to the general ledger in the format provided in the...
-
If inventory (opening) is $800, purchases $4000, freight inwards $100 and inventory (closing) $1000, what is the cost of sales?
-
Consider a new product or service that has recently become available for purchase by consumers. To what extent did this product or service possess the screening characteristics that are described in...
-
Do the three planes x + 2x + x 3 = 4, X X 3 = 1, and x + 3x = 0 have at least one common point of intersection? Explain.
-
At what position, and on what ring, would you expect bromination of benzanilide to occur? Explain by drawing resonance structures of theintermediates. Benzanilide 'N' H.
-
Would you expect the FriedelCrafts reaction of benzene with (R)-2-chloro- butane to yield optically active or racemic product explain.
-
How would you synthesize the following substances starting from benzene or phenol? Assume that ortho- and Para-substitution products can be separated. (a) o-Bromobenzoic acid (b) p-Methoxytoluene (c)...
-
An airline will purchase 2 million gallons of jet fuel in one month and hedges using heating oil futures. From historical data, the standard deviation of heating oil futures contract is sF =0.031...
-
A European call option for a share costs $5.00. The exercise price of the call option is $100.00. An investor buys one call and holds it until maturity: a. Under what circumstances will the holder of...
-
You have been asked to estimate the cost of capital for the UTX corporation. The company has 7 million shares and 150,000 bonds outstanding at par value $10,000. In addition, it has $300 million in...

Study smarter with the SolutionInn App


